Announcing the courses of the Computer Center at the College of Medicine
The Sheikh Abdullah Al-Nouri Charity Association in Kuwait donated one million four hundred thousand Egyptian pounds to support the Trauma Hospital at Assiut University.
Announcement of basic courses for appointment to the rank of assistant teacher or teacher - Measurement and Evaluation Center - August 2023
The Faculty of Medicine, Assiut University, participates in the medical curative convoy at the Martyr Ahmed Galal Official Language School

كلية الطب جامعة أسيوط تشارك في القافلة الطبية العلاجية بمدرسة الشهيد أحمد جلال الرسمية للغات
إستمرار للدور الرائد الذى تقدمه كلية الطب على الصعيدين التعليمى والخدمى لمجتمعها المحيط وما تحققه من جهد ملموس بقطاع خدمة المجتمع وتنمية البيئة لخدمة المواطنين المحتاجين وكبار السن.
إنطلقت فعاليات القوافل الطبية الخيرية العلاجيه لتوقيع الكشف الطبى والعلاج بالمجان بمدرسة الشهيد/ أحمد جلال الرسمية للغات بالتعاون مع مديرية الصحة بأسيوط
تحت رعاية
اللواء/ عصام سعد.- محافظ أسيوط
أ.د/ أحمد المنشاوى - رئيس الجامعة
وبحضور أ.د/ محمود عبد العليم- نائب رئيس الجامعة لشئون خدمة المجتمع وتنمية البيئة
ا.د/ محمد زين- وكيل وزارة الصحة والسكان بأسيوط
وبإشراف
أ.د/ علاء عطية- عميد الكلية
أ.د/ سعد زكى محمود- وكيل الكلية لشئون خدمة المجتمع وتنمية البيئة والمشرف العام على القوافل الطبية
د/ عمرو محمد محمد عبد المجيد - مشرف مركز القوافل الطبية
وبمشاركه كبار الاطباء للعمل بالقافله الطبيه من خمس تخصصات مختلفة وبحضور دكتور ميشيل ميلاد فكري و دكتور عبد الكريم واحمد عبد الجبار من الباطنه العامه و دكتور احمد عبد الفتاح من الجراحه العامه و دكتوره ايه احمد حسن يونس من جلديه وتناسليه ودكتوره رحاب محمد راشد ودكتوره ميرنا عزت نظير من طب الأطفال و دكتور احمد ايمن مصطفى من انف واذن وحنجره.
وتأتي القافله ضمن سلسله من القوافل الطبيه المتكامله التي تطلقها الكليه بشكل دوري ومستمر وتشارك بها ضمن مبادره رئاسه الجمهوريه "حياه كريمه" وتم توقيع الكشف الطبي على ١٢٠٠ حاله وتحويل ٣٨ حاله للمستشفيات الجامعيه لمتابعتها واتخاذ الإجراءات اللازمة.
Congratulations to the faculty members on the new promotions

تهنئ العلاقات العامة والإعلام بكلية الطب ووحدة الخدمات التكنولوجية السادة اعضاء هيئة التدريس بالترقيات الجديدة :
 تعيين الدكتورة / أمل محمد على تهامى _ لشغل وظيفة أستاذ بقسم الأمراض العصبية والنفسية
تعيين الدكتورة / أمل محمد على تهامى _ لشغل وظيفة أستاذ بقسم الأمراض العصبية والنفسية
 تعيين الدكتور/ هيثم على حسن على _ لشغل وظيفة أستاذ بقسم جراحة الأوعية الدموية
تعيين الدكتور/ هيثم على حسن على _ لشغل وظيفة أستاذ بقسم جراحة الأوعية الدموية
 تعيين الدكتورة / شيرين فرغلى جاد أحمد _ لشغل وظيفة أستاذ بقسم الأمراض الصدرية
تعيين الدكتورة / شيرين فرغلى جاد أحمد _ لشغل وظيفة أستاذ بقسم الأمراض الصدرية
 تعيين الدكتور / أحمد محمود فهمى فتح الله _ لشغل وظيفة أستاذ بقسم طب وجراحة العيون
تعيين الدكتور / أحمد محمود فهمى فتح الله _ لشغل وظيفة أستاذ بقسم طب وجراحة العيون
 تعيين الدكتورة / داليا محمد محمد على السبيتى _ لشغل وظيفة أستاذ بقسم طب وجراحة العيون
تعيين الدكتورة / داليا محمد محمد على السبيتى _ لشغل وظيفة أستاذ بقسم طب وجراحة العيون
 تعيين الدكتور / مجدى محمد مصطفى حسن _ لشغل وظيفة أستاذ مساعد بقسم طب وجراحة العيون
تعيين الدكتور / مجدى محمد مصطفى حسن _ لشغل وظيفة أستاذ مساعد بقسم طب وجراحة العيون
 تعيين الدكتورة / داليا محمد على تهامى _ لشغل وظيفة أستاذ مساعد بقسم طب وجراحة العيون
تعيين الدكتورة / داليا محمد على تهامى _ لشغل وظيفة أستاذ مساعد بقسم طب وجراحة العيون
 تعيين الدكتور / عمرو حسن ابراهيم أبو فدان _ لشغل وظيفة أستاذ مساعد بقسم جراحة المسالك البولية والتناسلية
تعيين الدكتور / عمرو حسن ابراهيم أبو فدان _ لشغل وظيفة أستاذ مساعد بقسم جراحة المسالك البولية والتناسلية
 تعيين الدكتور / أحمد البدرى إبراهيم أبو النور _ لشغل وظيفة أستاذ مساعد بقسم جراحة المسالك البولية والتناسلية
تعيين الدكتور / أحمد البدرى إبراهيم أبو النور _ لشغل وظيفة أستاذ مساعد بقسم جراحة المسالك البولية والتناسلية
 تعيين الدكتور / محمود ثابت أيوب _ لشغل وظيفة أستاذ مساعد بقسم الجراحة العامة
تعيين الدكتور / محمود ثابت أيوب _ لشغل وظيفة أستاذ مساعد بقسم الجراحة العامة
 تعيين الدكتورة/ ايمان رضا محمد بدوى- لشغل وظيفة استاذ مساعد بقسم الباثولوجيا الأكلينيكية
تعيين الدكتورة/ ايمان رضا محمد بدوى- لشغل وظيفة استاذ مساعد بقسم الباثولوجيا الأكلينيكية
 تعيين الدكتور / محمود عادل علي فيصل _ لشغل وظيفة مدرس بقسم التخدير والعناية المركزة وعلاج الآلم
تعيين الدكتور / محمود عادل علي فيصل _ لشغل وظيفة مدرس بقسم التخدير والعناية المركزة وعلاج الآلم
 تعيين الدكتورة/ سارة محمد منصور فتحى على _ لشغل وظيفة مدرس بقسم التخدير والعناية المركزة وعلاج الألم
تعيين الدكتورة/ سارة محمد منصور فتحى على _ لشغل وظيفة مدرس بقسم التخدير والعناية المركزة وعلاج الألم
 تعيين الدكتور / عبد الله رجب عبد الرحمن مرسى _ لشغل وظيفة مدرس بقسم جراحة المخ والأعصاب
تعيين الدكتور / عبد الله رجب عبد الرحمن مرسى _ لشغل وظيفة مدرس بقسم جراحة المخ والأعصاب
 تعيين الدكتور / ياسر عبد الموجود فيصل ابراهيم _ لشغل وظيفة مدرس بقسم الأمراض الباطنة
تعيين الدكتور / ياسر عبد الموجود فيصل ابراهيم _ لشغل وظيفة مدرس بقسم الأمراض الباطنة
 تعيين الدكتور / عمرو عصام سعد محمد _ لشغل وظيفة مدرس بقسم جراحة المسالك البولية والتناسلية
تعيين الدكتور / عمرو عصام سعد محمد _ لشغل وظيفة مدرس بقسم جراحة المسالك البولية والتناسلية
 تعيين الدكتورة/ شيماء كامل ابراهيم حادي _ لشغل وظيفة مدرس بقسم طب وجراحة العيون
تعيين الدكتورة/ شيماء كامل ابراهيم حادي _ لشغل وظيفة مدرس بقسم طب وجراحة العيون
 تعيين الدكتورة / نورهان طالب محمد _ لشغل وظيفة مدرس بقسم الأمراض الباطنة ( تخصص أمراض الدم الاكلينيكية وزع النخاع ) .
تعيين الدكتورة / نورهان طالب محمد _ لشغل وظيفة مدرس بقسم الأمراض الباطنة ( تخصص أمراض الدم الاكلينيكية وزع النخاع ) .
 تعيين الدكتورة / ايه عاطف فاروق محمد حسن القرن _ لشغل وظيفة مدرس بقسم الصحة العامة وطب المجتمع
تعيين الدكتورة / ايه عاطف فاروق محمد حسن القرن _ لشغل وظيفة مدرس بقسم الصحة العامة وطب المجتمع
 تعيين الدكتور / عمرو عبدالله عبد المعبود محمد مهران _ لشغل وظيفة مدرس بقسم جراحة المسالك البولية والتناسلية
تعيين الدكتور / عمرو عبدالله عبد المعبود محمد مهران _ لشغل وظيفة مدرس بقسم جراحة المسالك البولية والتناسلية
 تعيين الطبيبة / رؤى المعتز عبد الرازق ذهنى _ لشغل وظيفة مدرس مساعد بقسم الأمراض العصبية والنفسية
تعيين الطبيبة / رؤى المعتز عبد الرازق ذهنى _ لشغل وظيفة مدرس مساعد بقسم الأمراض العصبية والنفسية
The success of a medical team in the Department of Diagnostic and Interventional Radiology and Arterial Catheterization in closing a large aneurysm in the basilar artery of the brain, which is known to be difficult to deal with.
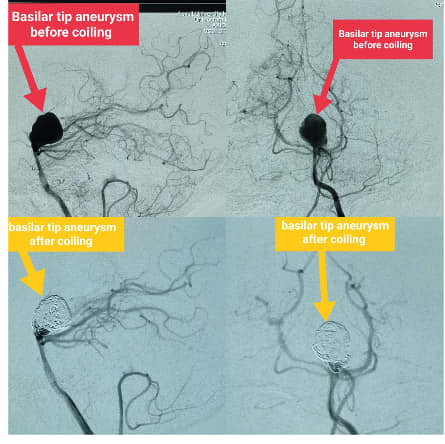
نجح فريق طبي بقسم الاشعة التشخيصية والتداخليه وقسطرة الشرايين في غلق تمدد دموي كبير بالشريان القاعدي للمخ والمعروف بصعوبة التعامل معه.
Coiling of large basilar tip aneurysm
وحول تفاصيل العملية فقد كشف الدكتور مصطفى هاشم الاستاذ بالقسم ورئيس القسم السابق ان المريض يبلغ من العمر ٥٥ عام وقد توجه إلى المستشفى يعاني من صداع شديد حاد ومستمر وقيء و بعد عمل أشعة مقطعية بالصبغة علي شرايين المخ وبعض الفحوصات و التحاليل اللازمة تم تشخيص الحاله علي انها تمدد منفجر ف الشريان القاعدي .
تم إنهاء إجراءات التأمين الصحي و عمل دخول للمريض بالقسم الخاص بالمستشفيات الجامعيه وتجهيزه لغرفة العمليات لعمل قسطرة مخية وتحت تخدير كلي عن طريق الشريان الفخذى ثم الوصول للشريان الفقرى الايسر وتم وضع ٤ ملفات حلزونية و٣ ثلاثية الابعاد ذاتيه الانفصال.
تم عمل قسطرة بعد انتهاء تركيب الملفات للتأكد من الغلق التام وعدم تأثر اي شريان من الشرايين المتجهه للمخ والمخيخ ثم وضع المريض بغرفة الافاقة بوحدة القسطرة وبعد تعافيه تم نقله للعناية المركزة المجهزة مسبقا فقط للمتابعة وبدون اي مضاعفات والمريض بحالة جيدة ومستقرة وبفضل الله وأطبائنا الكرام تم شفاء المريض وعاد الي منزله سليم معافي .
تمت القسطرة المخيه بقيادة الاستاذ الدكتور مصطفي هاشم استاذ الاشعة التشخيصية والتداخلية ومن المدرسين الدكتور محمود رفعت والدكتور حمدى محمد ابراهيم والمدرس المساعد الطبيب عمر جمال والطبيب مقيم هاجر لطف الله وإشراف طاقم التخدير بالمستشفى الدكتور عمر حسن وبمساعدة من طاقم مساعد متميز من التمريض والفنيين والمساعدين .
وتوجه الفريق الطبي بالشكر للأستاذ الدكتور مصطفى الشرقاوي رئيس قسم الأشعة التشخيصية والتداخلية والاستاذ الدكتور عصام مناع رئيس قسم التخدير والعنايه المركزه
وتلك العملية من العمليات الصعبة التى تدل على مهارة ودقة الكادر الطبى المسئول عن العملية ، وتعكس الطفرات الكبرى التى يشهدها القطاع الطبى بالمستشفيات الجامعية.
والجدير بالذكر أن وحدة قسطرة الشرايين بقسم الاشعه عريقة منذ نهاية الثمانينات وقامت بإجراء ما يقرب من ١٠٠ حالة قسطرة مخية علاجيه في السنوات الاخيره ما بين تمدد دموي لشرايين المخ وناسور شرياني وريدي بالمخ وغلق تشوهات الأوعية الدموية للمخ وشفط جلطات حادة لشرايين المخ وتركيب دعامات للشريان السباتي المغذى للمخ وغلق الاوعيه المغذيه للاورام ماقبل إجراء العمليات وعلي صعيد آخر أجرت مئات الحالات التشخيصية لشرايين المخ.
وتضم الوحدة ٤ اساتذة واستاذ مساعد و ٦ مدرسين بالإضافة إلى الهيئة المعاونة من مدرسين مساعدين ونواب.
ولا يقتصر عمل الوحدة علي القسطرة المخية فقط ولكن يمتد لقسطرة الكبد والرئة والكلي والنزيف الدموي الحاد للاجزاء المختلفة من الجسم من قيء دموي ونزيف ناتج عن الإصابات والحوادث وغيرها وذلك علي مدار ٢٤ ساعة للطوارئ وقوائم يومية للحالات المستقرة بوحدتي القسطره بمستشفى الراجحي وكذلك المستشفي الرئيسي.
Association of serum fentanyl concentration and oxygen desaturation with oversedation in neonates
Fentanyl is an intravenously administered sedative during tracheal intubation in neonates with respiratory distress. Overdose of fentanyl leads to oversedation, as there is a direct concentration-effect relationship between the fentanyl serum concentration and respiratory depression in adults [1]. However, the relationship between serum fentanyl concentration and oxygen desaturation with oversedation in neonates still unclear. Our aim is to evaluate the association between serum fentanyl concentrations and oxygen desaturation with oversedation in neonates receiving artificial ventilation. Patients Our retrospective study included neonate patients given fentanyl for sedation during tracheal intubation. The patients were considered eligible if we could obtain the serum sample during the fentanyl therapeutic period. Measurement of serum fentanyl concentrations Blood samples were collected from the neonates as a routine care. Serum fentanyl concentrations were measured by liquid chromatography-tandem mass spectrometry. Oxygen desaturation Oxygen desaturation (ODS) was defined as percutaneous oxygen saturation less than 90%. During the fentanyl therapeutic period, All serum samples were collected during this period (the points with the highest concentrations were adopted) The occurrence of ODS (%SpO2<90%) was observed. Bayesian estimation Bayesian estimations were performed using the MwPharm++ software. The population pharmacokinetic parameters for fentanyl were established based on a previous report [2]. Volume of distribution (Vd)= 5.26 L/Kg Clearance (CL)= 3.6 L/h. There were no significant differences in patient characteristics between patients with and without ODS. The median fentanyl initial dose tended to be lower in patients with ODS compared to non-ODS patients. There was no significant difference in fentanyl concentration between patients with and without ODS. The fentanyl concentration per dose (C/D) in patients with ODS was significantly higher than in those with non-ODS. Time to incidence of ODS had a trend to be shorter than the entire fentanyl therapeutic period in the patients with non-ODS. Exposure time may not contribute to the incidence of ODS.
Development of Physiologically Based Pharmacokinetic Model of Risperidone and Extrapolation to Different Ethnic Groups and Special Population
Purpose: Physiologically based pharmacokinetic (PBPK) analyses have been frequently used in the clinical pharmacology section of regulatory applications. However, whether models developed and validated in healthy populations can be extrapolated to untested populations is not well known. This study aimed to determine whether a drug-specific PBPK model validated in a healthy population could be used to predict drug disposition in populations with different ethnicities, ages, genetic phenotypes, and pregnant population considering an example for risperidone and its active metabolite, paliperidone. Methods: PBPK modeling and simulation were performed using Simcyp Population-based ADME Simulator version 20. The risperidone and paliperidone compound models were developed based on physicochemical and pharmacokinetic parameters reported in the literature. The model was validated using observed values from 25 clinical studies including 15 in the adult population (8 Caucasian, 5 Chinese, and 2 Japanese), 8 in the pediatric population (5 Caucasian, 2 Chinese, and 1 Japanese), and 2 in the pregnant population. Visual predictive checks (VPC) for predicted and observed plasma concentrations, goodness-of-fit plots, prediction fold error, mean error (ME), and root-mean-square error (RMSE) were used for the graphical and statistical analyses. Results: Almost all (98.9%) of observed serum risperidone and paliperidone concentrations werewithin the 90% prediction intervals of each concentration by the PBPK simulation. All predicted values of serum risperidone concentration and AUC values in adult, pediatric, and pregnant populations met the 2fold acceptance criterion. A 91.8% of predicted values of paliperidone concentration and 97.4% of AUC values fall within the 2-fold criterion in all populations. The mean error (ME) and root-mean-square error (RMSE) for all predicted Cmax for risperidone were 0.61±0.4%, and 2.85%, respectively. Regarding paliperidone, the values were -0.038±0.45% and 3.16%, respectively. Conclusion: This study successfully shows an experimental application of PBPK modeling for the adaptation of ethnic variety and pediatric population.
Development of a physiologically based pharmacokinetic model of fentanyl for preventing central toxicity in neonate patients on respiratory support
Background: Fentanyl is widely used for sedation in preterm neonates requiring artificial ventilation. Neonates are especially vulnerable to side effects such as respiratory depression with oversedation which has a direct concentration-effect relation. The dosage regimen is usually derived from adults employing an extrapolation based on body weight which cannot accurately determine organ maturation in neonates. There is a necessity for model-based precision dosing of fentanyl that includes maturation. This study aimed to develop a pragmatic physiologically based pharmacokinetic (PBPK) model of fentanyl and to evaluate its prediction accuracy in neonates. Methods: PBPK modeling and simulation were performed using SimCYP Population-based ADME Simulator version 20. The fentanyl compound model was developed based on physicochemical and pharmacokinetic parameters reported in the literature. The model was validated using observed values from 25 clinical studies on adults and 4 on pediatrics. Serum samples of 14 neonates were collected as routine care. Serum fentanyl concentration was measured by liquid chromatography-mass spectrometry. Results: 100% of the predicted values of serum fentanyl concentration in adult and pediatric patients met the 2-fold acceptance criterion. Mean error (ME) and root-mean-square error (RMSE) for all predicted values were 0.28±0.15%, and 1.16%, respectively. In our neonatal patients, 95.5% of the predicted values were within the 2-fold acceptance criterion with ME and RMSE of 0.05±0.075%, and 0.34%, respectively. Median predicted fentanyl AUC value in patients with severe oxygen desaturation (sODS) (percutaneous oxygen saturation <80%) was higher than non- or moderate- ODS (nmODS) patients (ODS: 213 ng/mL·h, nmODS:25.2 ng/mL·h, P=0.043), while no significant difference was observed in fentanyl clearance depending on ODS (sODS: 1.33 L/h, nmODS: 1.39 L/h, P=0.35). Conclusion: This study shows that PBPK modeling can provide a precise prediction of serum fentanyl concentration in neonates. This model may be a useful tool for dose optimization and individualization strategies to avoid oversedation.
 Do you have any questions?
Do you have any questions?