Outcome predictability for obstetric conditions requiring intensive care admission”
Nesfatin-1, Dopamine, and NADPH levels in Infertile Women with Polycystic Ovary Syndrome: Is There a Relationship Between Their Levels and Metabolic and Hormonal Variables
Effects of anesthetic management on inflammatory markers in patients after major abdominal surgeries: A double blind controlled study
BACKGROUND: Surgical trauma induces systemic inflammatory response due to hormonal, immunological, and metabolic mediators associated with augmented secretion of various stress hormones. Opioids and local anesthetics might suppress Hypothalamic-pituitary-adrenal (HPA) axis function, probably due to their analgesic effect, suppressing stress hormone production. We aimed to evaluate the influence of different analgesic models on postoperative pain and inflammatory markers modulation after major abdominal surgeries.
METHODS: A total of 105 patients scheduled for elective abdominal colorectal surgeries were selected. Patients received 15 mg Bupivacaine intrathecal (IT) followed by general anesthesia after stabilizing the block level at T-4. They received fluids infusion during the operation and for four hours after. They were randomly assigned to one of three study groups: Group-1 (GM): received four microgram/kg IT morphine, Group-2 (GML): received four microgram/kg IT morphine plus 1.5 mg/kg intravenous Lidocaine loading dose then 2 mg/min with the saline infusion during the operation and the next 4 hours postoperative and Group-3 (GC): (control group) received no added drugs
RESULTS: The mean pain score was significantly (P<0.001) lower in the GML group than the other groups. GML group showed the lowest level of tumor necrosis factor-alpha (P <0.001;) followed by GM and control groups (10.3 ± 4.4 vs. 20 ± 4.4 vs. 26 ± 7.5, respectively). The level of Transforming Growth Factor beta 1 was significantly (P<0.001) higher in GML, followed by GM, and then GC (43.1 ± 12.5, 26 ± 4.2, and 18.9 ± 7.7, respectively). Opioid consumption was significantly (P<0.001) lower in GML than the other two groups.
CONCLUSION: Intraoperative and early postoperative intravenous lidocaine infusion significantly improved the quality of postoperative analgesia. Optimizing analgesia in anesthetic management has a favorable effect on the pro and anti-inflammatory mediators.
Keywords: Abdominal surgeries, Postoperative pain, TNF-alpha and TGF-beta
Coupling size exclusion chromatography to ultracentrifugation improves detection of exosomal proteins from human plasma by LC-MS
Diabetes Mellitus under the age of one year :clinical pattern, etiological factorsand possible mutation in KCNJ11 gene encoding Adenosine Triphosphate sensitive potassium …
Announcement regarding newly purchased books in the library
Seminar - Dr. Esraa Khaled Mohamed Nafady - Department of Anatomy
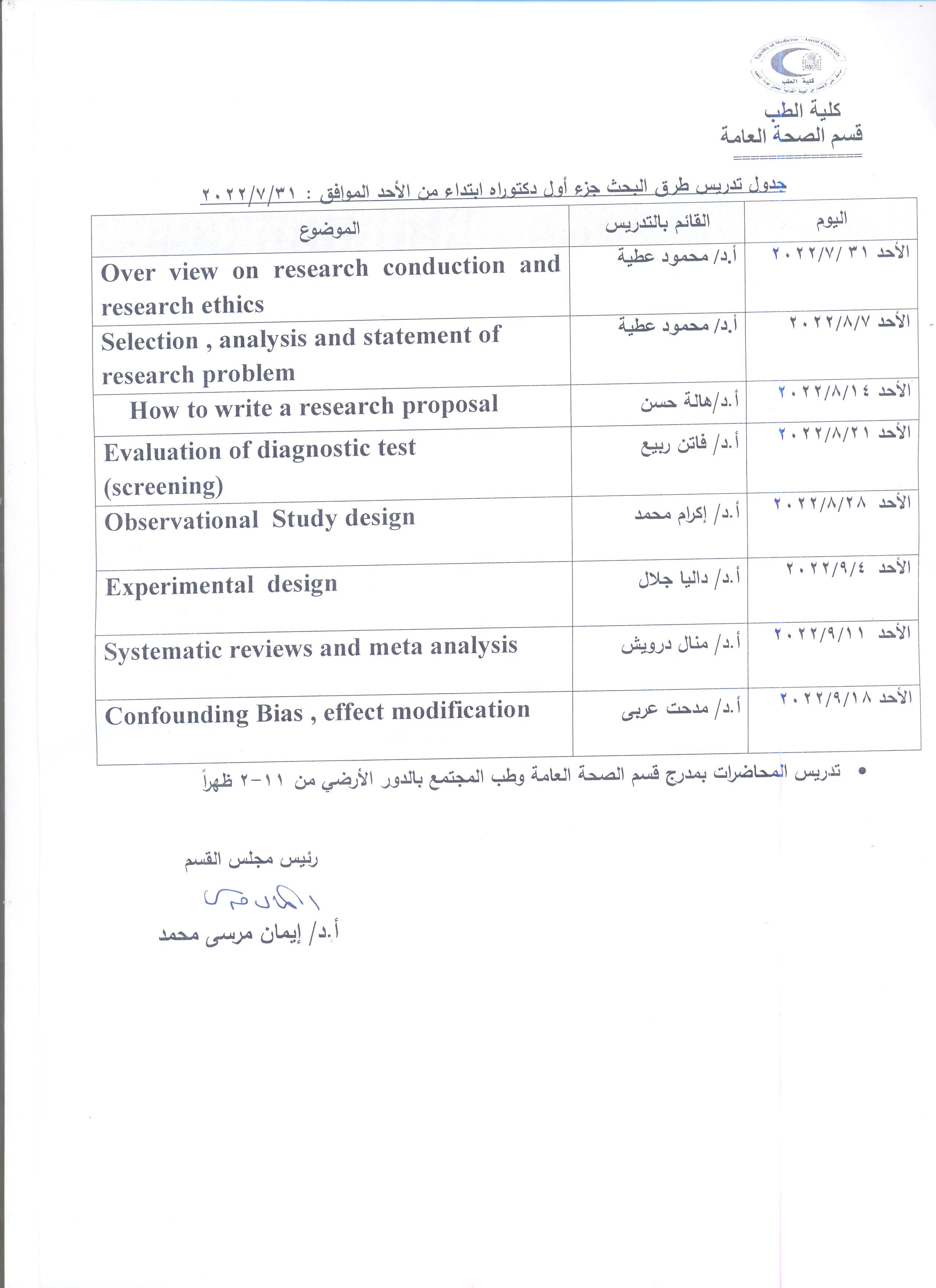
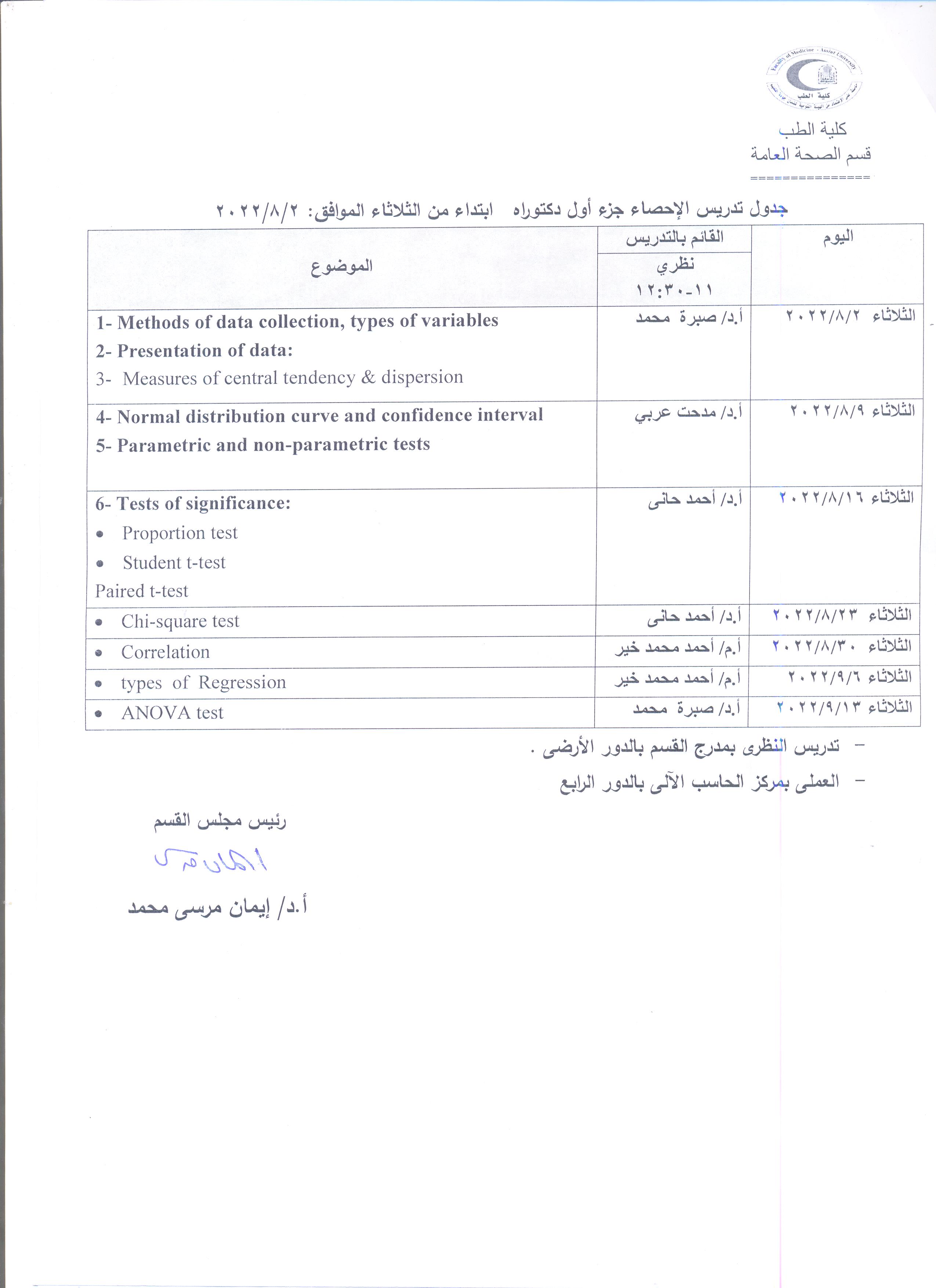
Announcement - Regarding the start of lectures for the first part of the Ph.D. starting from Sunday 31-7-2022
Advancing combination treatment with cilostazol and caffeine for Alzheimer’s disease in high fat-high fructose-STZ induced model of amnesia
Several studies have suggested that phosphodiesterase (PDE) inhibitors may be a disease-modifying for Alzheimer’s disease (AD). Cilostazol (CSZ) has been shown to be a new treatment for cognitive impairment with
limited efficacy. Our aim was to investigate the effect of caffeine on the efficacy of CSZ against STZ-induced type
2 diabetes (T2D)-related cognitive impairment in high fat/high fructose fed rats. The efficacy of low doses of
caffeine, CSZ, and CSZ plus caffeine against abnormal behavioral, biochemical, histological, or genetic changes of animal models of AD was examined. Eight weeks treatment with CSZ plus caffeine was more effective than CSZ or caffeine in improving impaired behavioral tests for cognition and memory. Histological examination exhibited a significant augmentation in the efficacy of CSZ by caffeine in protecting neurons from damage in T2D rats. Importantly, CSZ and caffeine normalized the accumulation of Amyloid beta (Aβ-42) and phosphorylated tau
protein (p-tau) positive cells in the brain of T2D rats. CSZ or CSZ plus caffeine reversed low glutamate gene
expression, elevated cholinesterase level, and elevated caspase-3 activity in T2D rats. Furthermore, CSZ plus
caffeine was significantly more effective than CSZ or caffeine in inhibiting the increase in malondialdehyde
(MDA) level, total oxidative stress, pro-inflammatory cytokines and glucogen synthase kinase-3 beta (GSK-3β) in
the hippocampus of T2D rats. Also, CSZ plus caffeine was more effective than CSZ or caffeine in alleviating
insulin resistance and hypercholesterolemia in T2D rats. Our findings suggest the possibility of effective treatment of AD by enhancing the therapeutic potential of CSZ through combined treatment with lower doses of
caffeine. The enhancement of CSZ effect by caffeine is attributed to the increased inhibitory effect of CSZ on
insulin resistance, GSK-3β activity, hypercholesterolemia, oxidative stress and pro-inflammatory cytokines.
 Do you have any questions?
Do you have any questions?