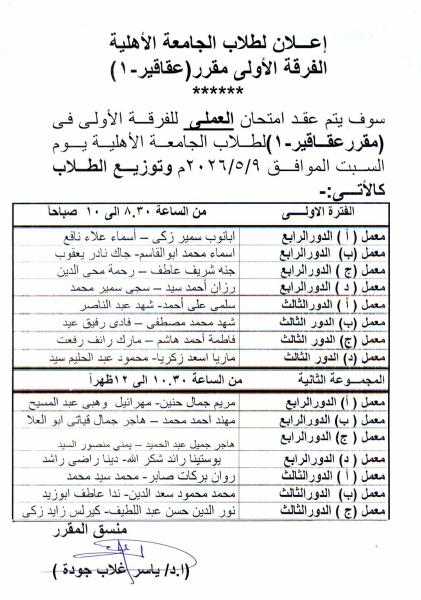
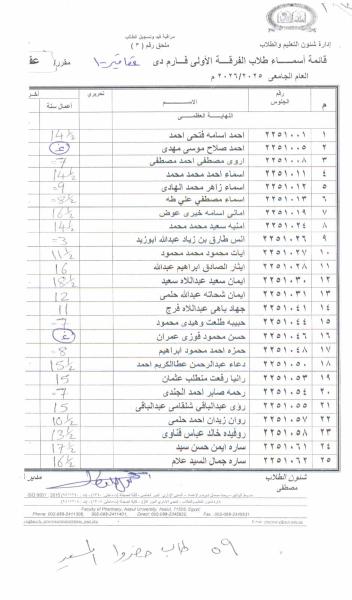
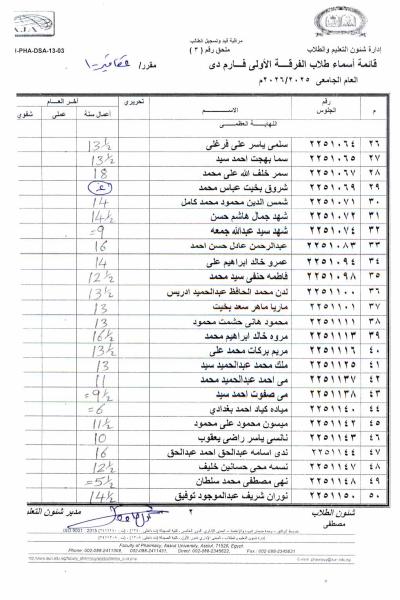
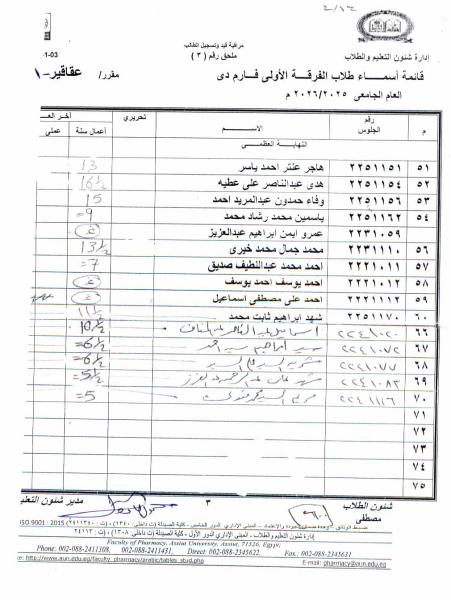
First: Medicinal Chemistry II (3rd Year – National University)
- Prof. Dr. Tarek Aboul-Fadl Mohammad
- Prof. Dr. Nawal Abou Bakr Abdel-Haleem El-Koussi
- Prof. Dr. Ahmed Safwat Aboraia
- Dr. Shorok Ahmad Mohammad Abdelhameed (Practical Supervisor)
Second: Medicinal Chemistry II (3rd Year – Pharm D)
- Prof. Dr. Farghaly Abdul Hamid Omar
- Prof. Dr. Nawal Abou Bakr Abdel-Haleem El-Koussi
- Dr. Shorok Ahmad Mohammad Abdelhameed (Practical Supervisor)
Third: Medicinal Chemistry II (3rd Year – Clinical Pharmacy)
- Prof. Dr. Tarek Aboul-Fadl Mohammad
- Prof. Dr. Nawal Abou Bakr Abdel-Haleem El-Koussi
- Prof. Dr. Ahmed Safwat Aboraia
- Dr. Shorok Ahmad Mohammad Abdelhameed (Practical Supervisor)
Fourth: Drug Design (4th Year – Pharm D)
- Prof. Dr. Tarek Aboul-Fadl Mohammad
- Dr. Shorok Ahmad Mohammad Abdelhameed (Practical Supervisor)
Fifth: Drug Design (Elective) – 5th Year Pharm D
- Prof. Dr. Tarek Aboul-Fadl Mohammad
- Prof. Dr. Ahmed Safwat Aboraia (Practical Supervisor)
Sixth: Drug Design (Elective) – Clinical Pharmacy
- Prof. Dr. Tarek Aboul-Fadl Mohammad
- Dr. Shorok Ahmad Mohammad Abdelhameed (Practical Supervisor)
Seventh: Scientific Writing (Level 6) – Clinical Pharmacy & National University
- Prof. Dr. Tarek Aboul-Fadl Mohammad
- Prof. Dr. Ahmed Safwat Aboraia
![]()