Do you have any questions? (088) 2080369 - 2345622 Pharmacy_QAAU@pharm.aun.edu.eg
Do you have any questions? (088) 2080369 - 2345622 Pharmacy_QAAU@pharm.aun.edu.eg
A novel route for fabrication of yellow emissive carbon dots for selective and sensitive detection of vitamin B12
Design and preparation of a fluorescent molecularly imprinted membrane for the selective detection of pepsin enzyme as a biomarker for gastroesophageal reflux disease.
Final Exam schedule for the second semester of the academic year 2024/2025 (Pharm D Clinical Pharmacy Program)
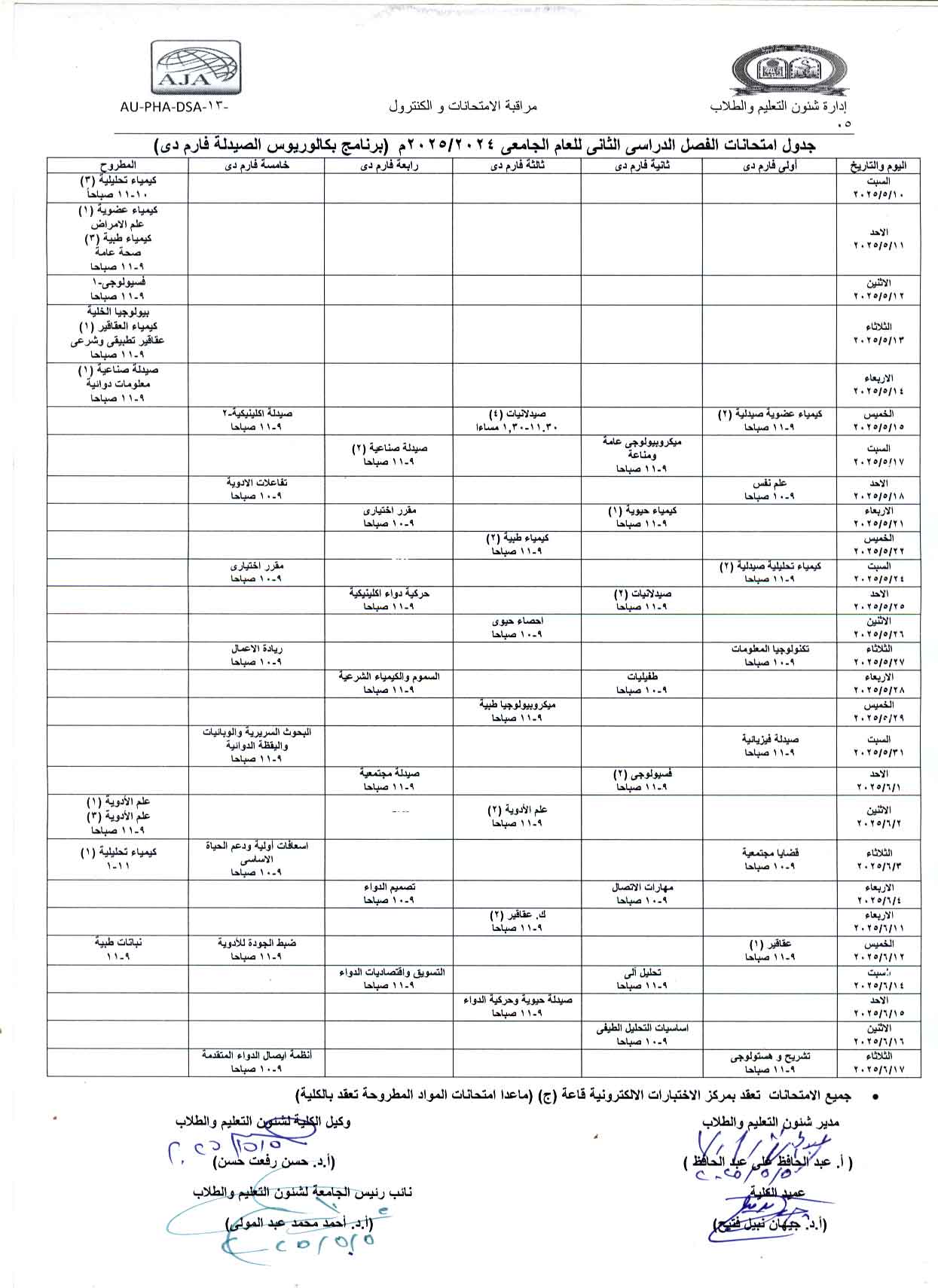
Final Exam schedule for the second semester of the academic year 2024/2025 (Pharm-D Bachelor of Pharmacy Program)
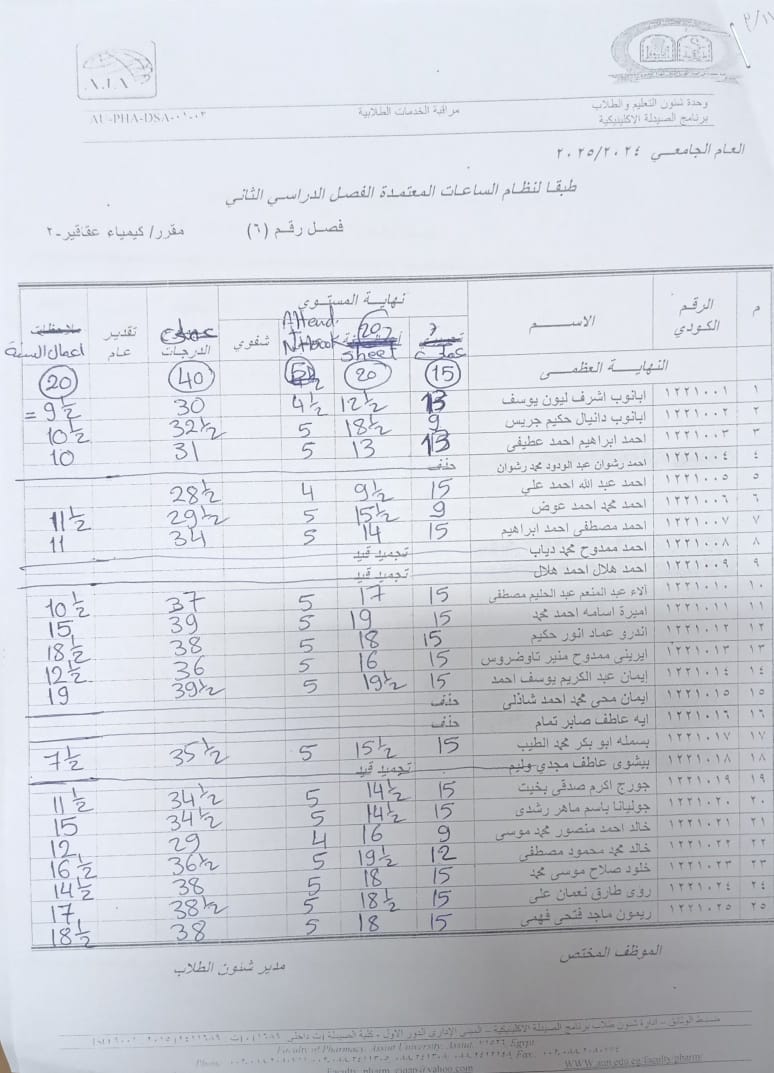
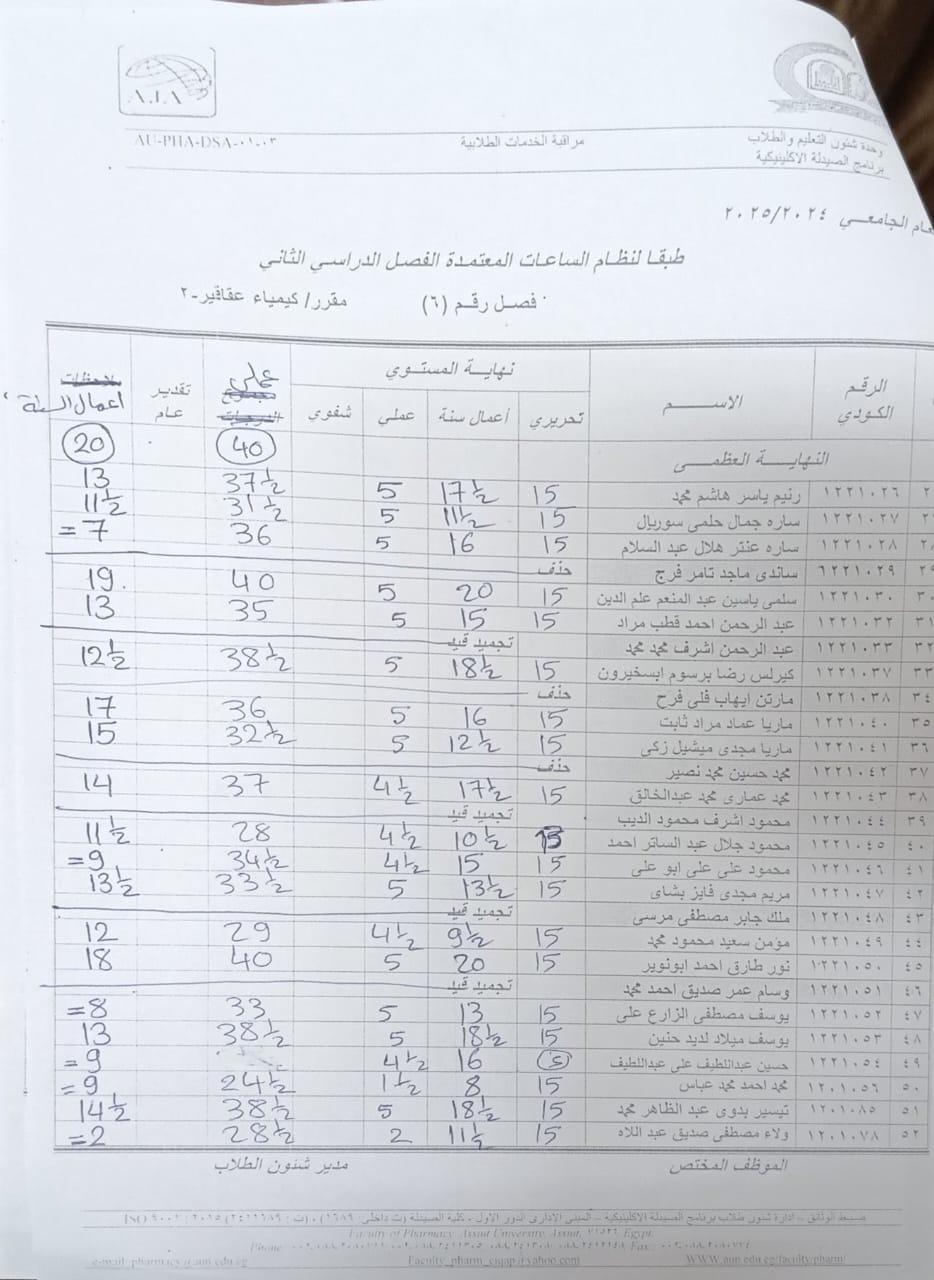
Announcement for Clinical Pharmacy Program Students – Class (6) Course: Phytochemistry II


The annual scientific conference of the faculty for the academic year 2024/2025

God willing, the
Faculty's annual scientific conference for the academic year 2024/2025 will be held
to discuss topics related to educational and research affairs in light of the recommendations of the departments' scientific conferences. This will be on Monday, May 5, 2025, at 12:00 PM, in the celebration hall in Building (B).

Meeting of the Council of the Pharmaceutics Department, Faculty of Pharmacy this is on The event will take place on Monday, May 5, 2025.

God willing, the meeting of the Pharmaceutics Department Board of the Faculty of Pharmacy No. (539) will be held on Monday, May 5, 2025 at ten o’clock in the morning
in the department board on the third floor under the chairmanship of the Faculty to discuss the topics that we will inform you later.
Meeting of the Council of the Medicinal Chemistry Department, Faculty of Pharmacy That will be Sunday, May 4, 2025

God willing, The Medicinal Chemistry Department Council will hold its regular monthly meeting number (485) That will be on Sunday, May 4, 2025, at 11:00 AM
In The Department Library
Meeting of the Council of the Pharmaceutical Analytical Chemistry Department, Faculty of Pharmacy This will take place on Sunday, May 4, 2025.

God willing, The Pharmaceutical Analytical Chemistry Department Council will hold its regular monthly meeting number (526) This will take place on Sunday, May 4, 2025, at 11:00 AM.