Do you have any questions? (088) 2080369 - 2345622 Pharmacy_QAAU@pharm.aun.edu.eg
Do you have any questions? (088) 2080369 - 2345622 Pharmacy_QAAU@pharm.aun.edu.eg
The exam date has been changed for the course s“Physiology and Analytical Pharmaceutical Chemistry-3” for the first semester of the academic year 2022/2021 AD - second year Pharm D
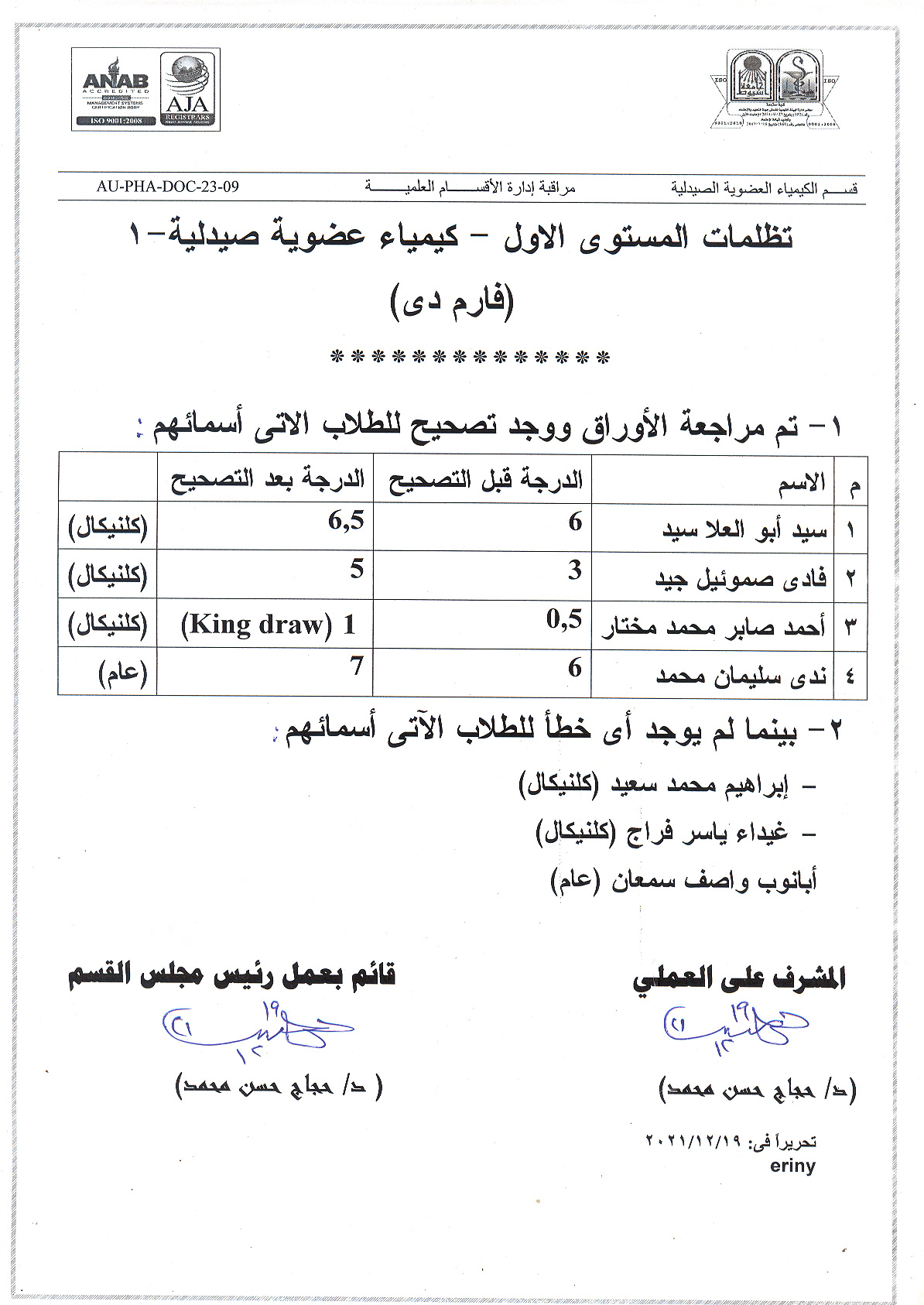
Pharmaceutical Organic Chemistry-1 (Pharm D) mid term results after grievance

Important Announcement for Semester-3 Students of the Pharm D Programs (Pharmacy and Clinical Pharmacy)


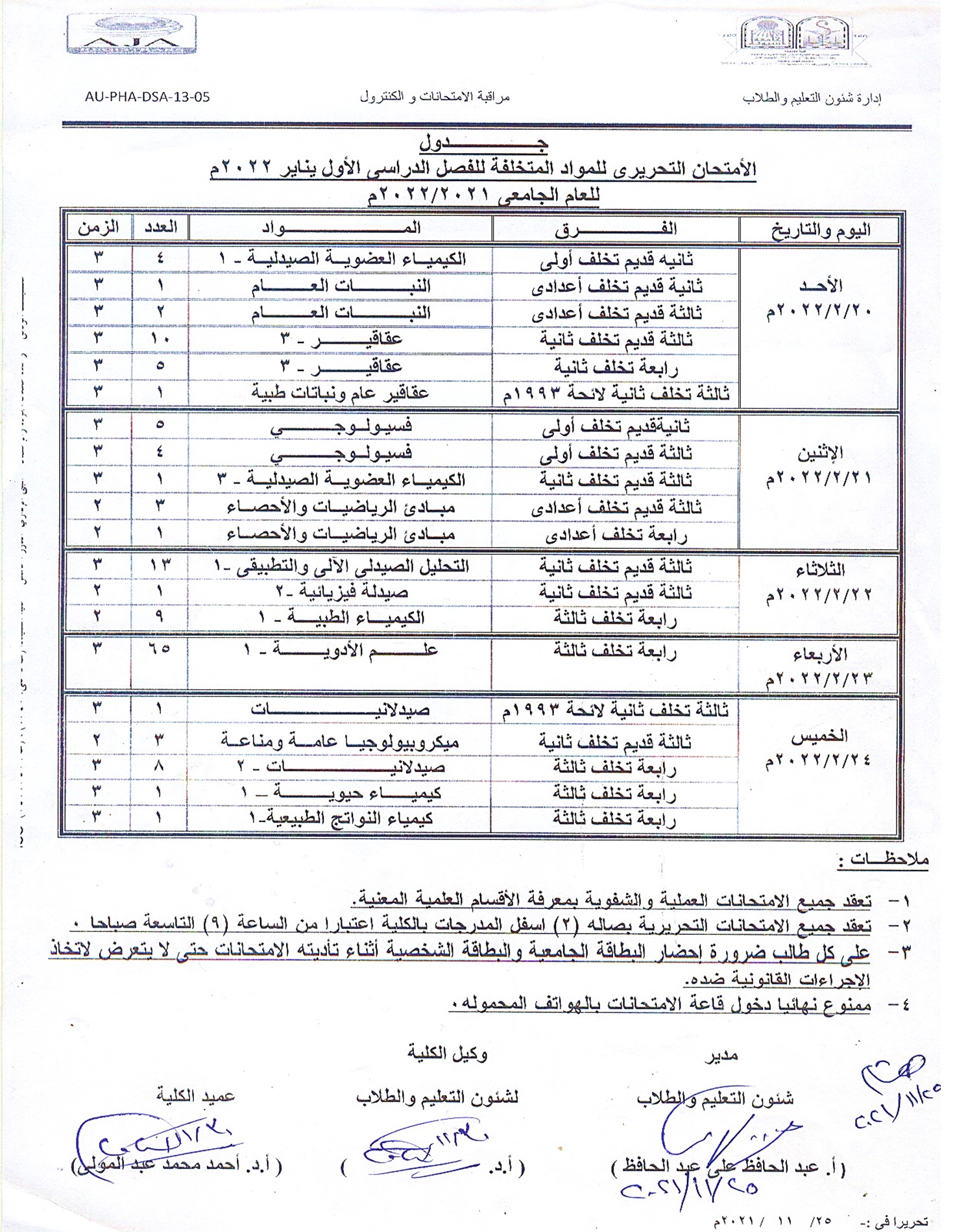
Schedule of the exams for the failed students for the first semester, January 2022, for the academic year 2021/2022
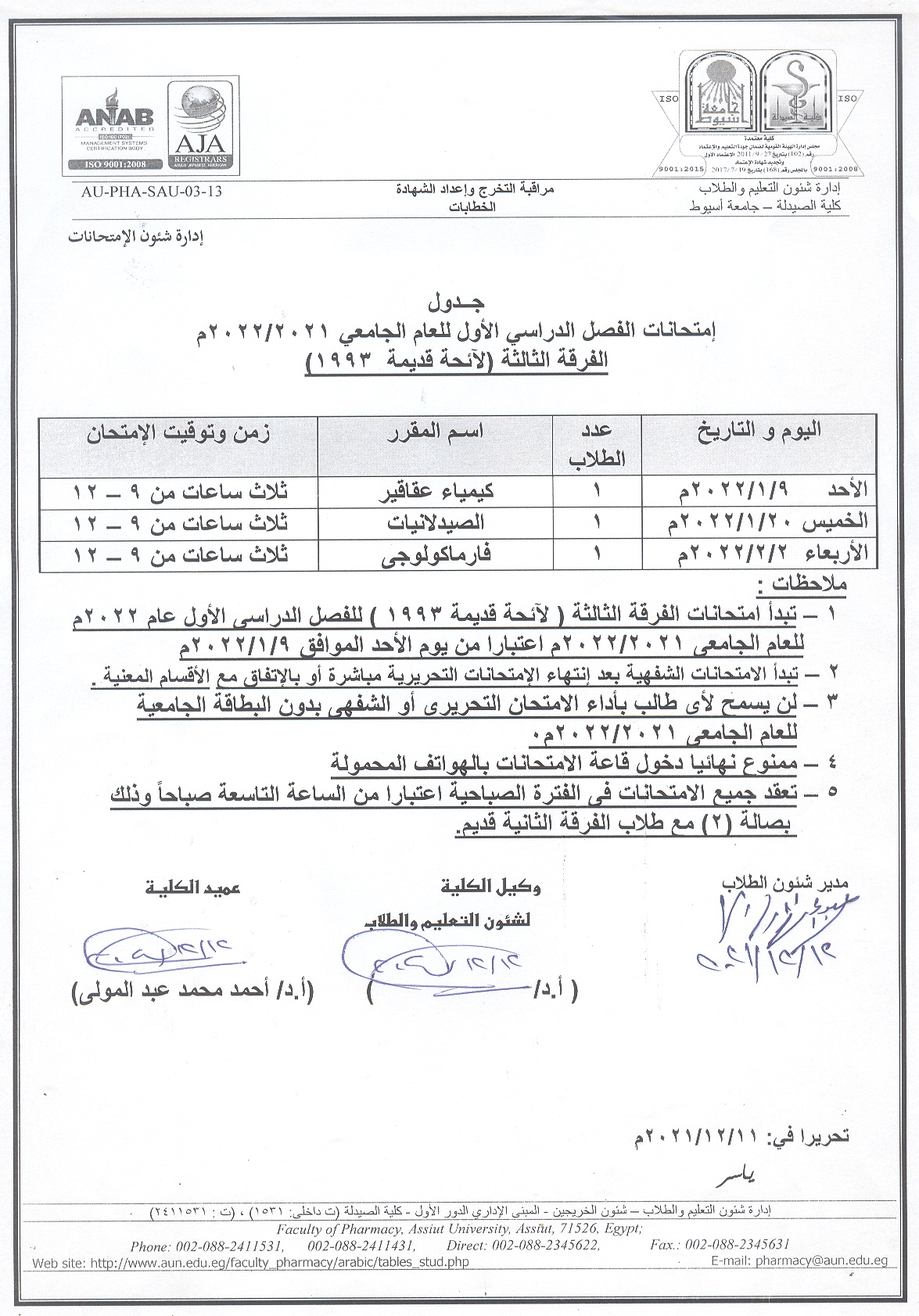
• Exam schedule for the first semester of the academic year 2021/2022 , the third year (old list 1993)
• Schedule of exams for students of the Clinical Pharmacy Program, first semester, for the academic year 2021/2022
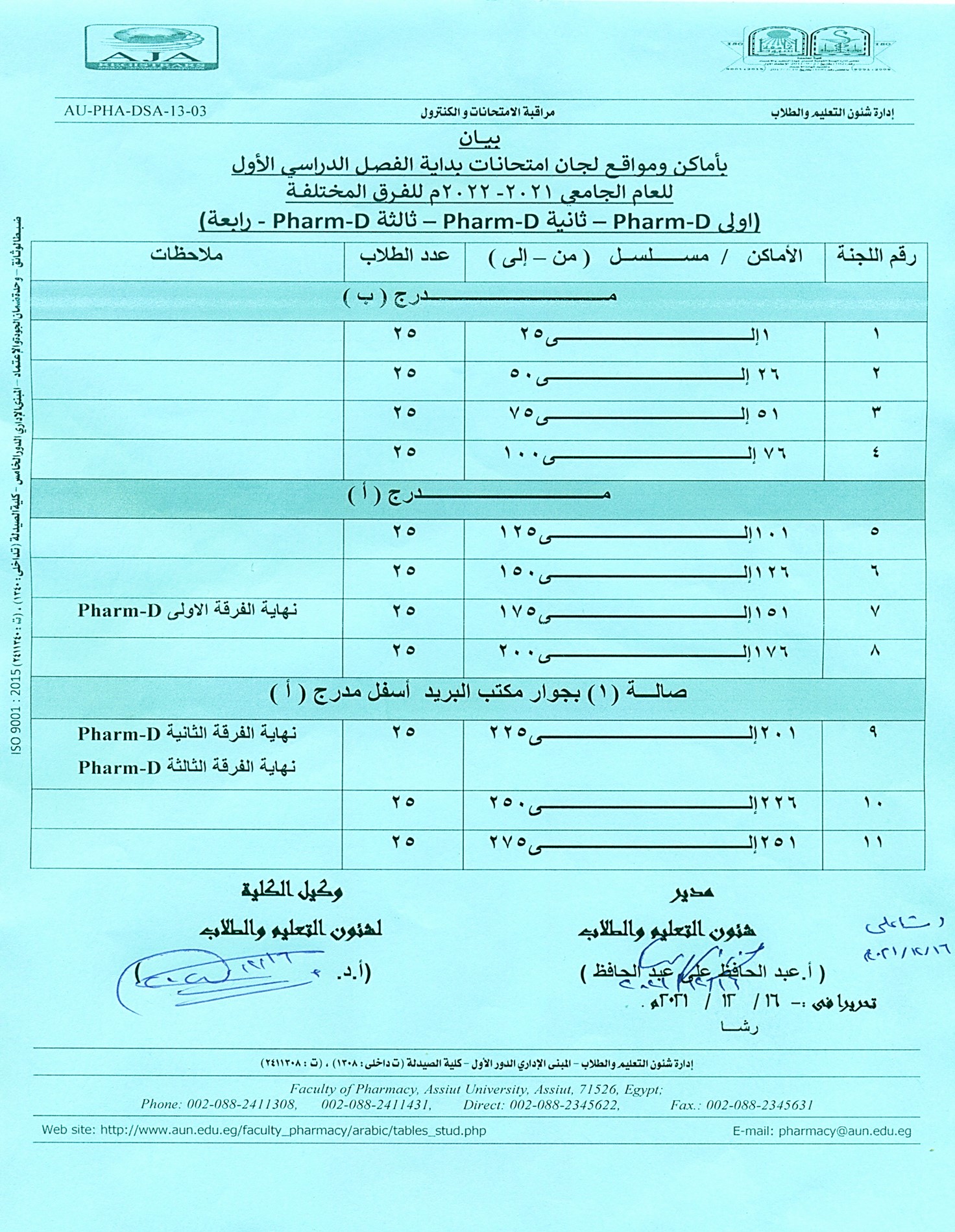
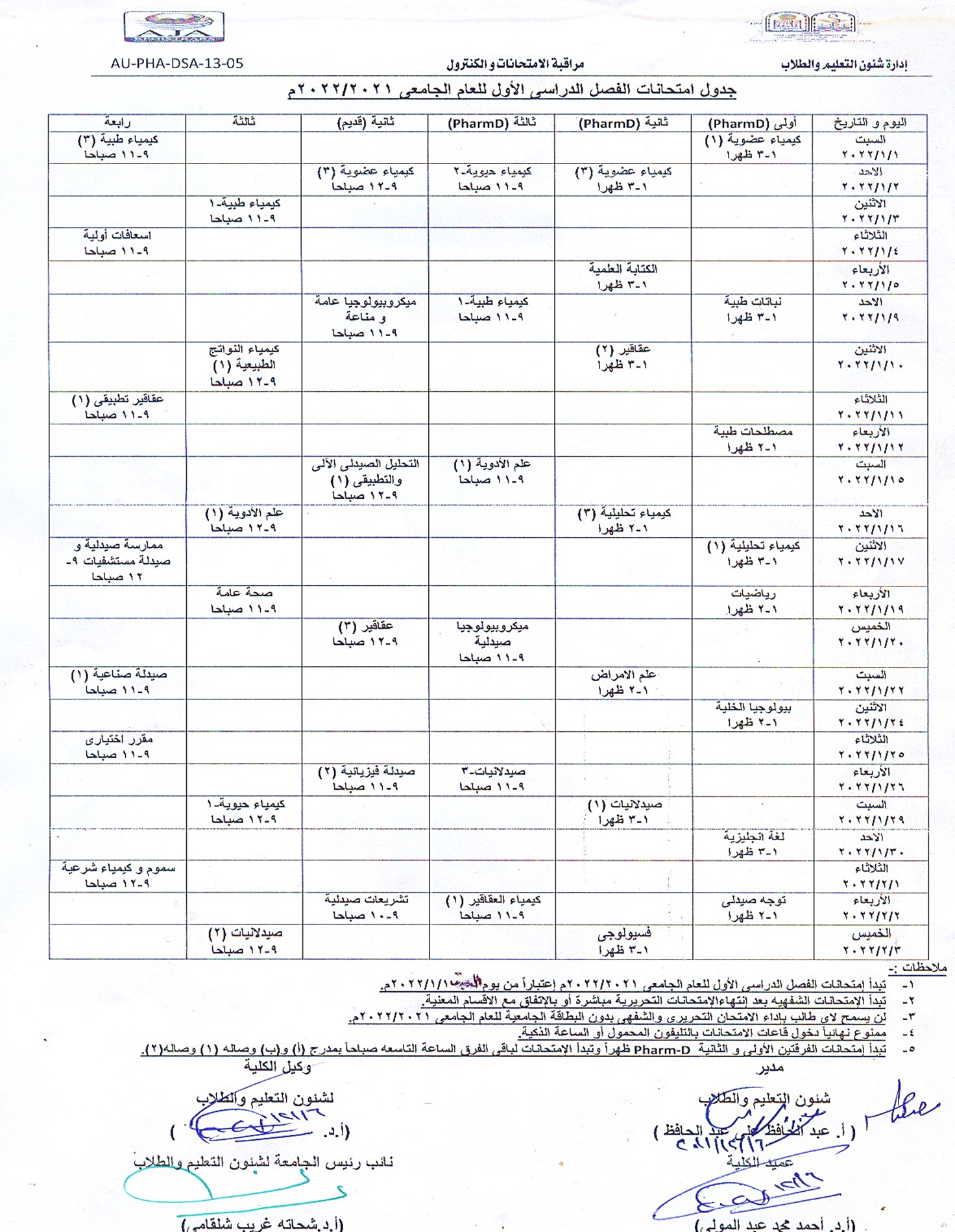
Exam schedule for the first semester of the academic year 2021/2022


Synthesis and anti-inflammatory testing of some new compounds incorporating 5 –aminosalicylic acid (5-ASA) as potential prodrugs
This work includes the synthesis of 15 final compounds (6a-h and 7b-h) as prodrugs of 5-ASA in the form of the acid itself, esters and amides linked by an amide linkage through a spacer to the endocyclic ring N of nicotinamide. Also, 15 new intermediate compounds were prepared. The target compounds (6b, 6f, 7b, and 7e-h) revealed potent analgesic and anti-inflammatory activities in comparison to sulfasalazine and 5-ASA. In addition, ulcerogenicity, LD50, in vivo and in vitro metabolism of compound 7f were determined.
Synthesis of trigonelline and nicotinamide linked prodrugs of 5 –aminosalicylic acid (5-ASA) with analgesic and anti-inflammatory effects
3-N-(4`-Hydroxy-3`-substituted phenyl)carbamoyl-1-methylpyridinium iodides (compds. 5b-j) and 3-carbamoyl-1-(N-(4`-hydroxy- 3`-substituted phenyl)carbamoyl) methyl pyridinium chlorides (compds. 7a-j) were synthesised and some of them were tested for their analgesic and antiinflammatory activities by hot plate test and carageenin-induced hind paw edema model, respectively. Compound 5b revealed the most potent analgesic and anti-inflammatory activities in comparison to sulfasalazine (SASP) and 5-ASA. In addition, ulcerogenicity, LD50, in-vivo and in vitro cleavage and pH stability of compound 5b were also determined.