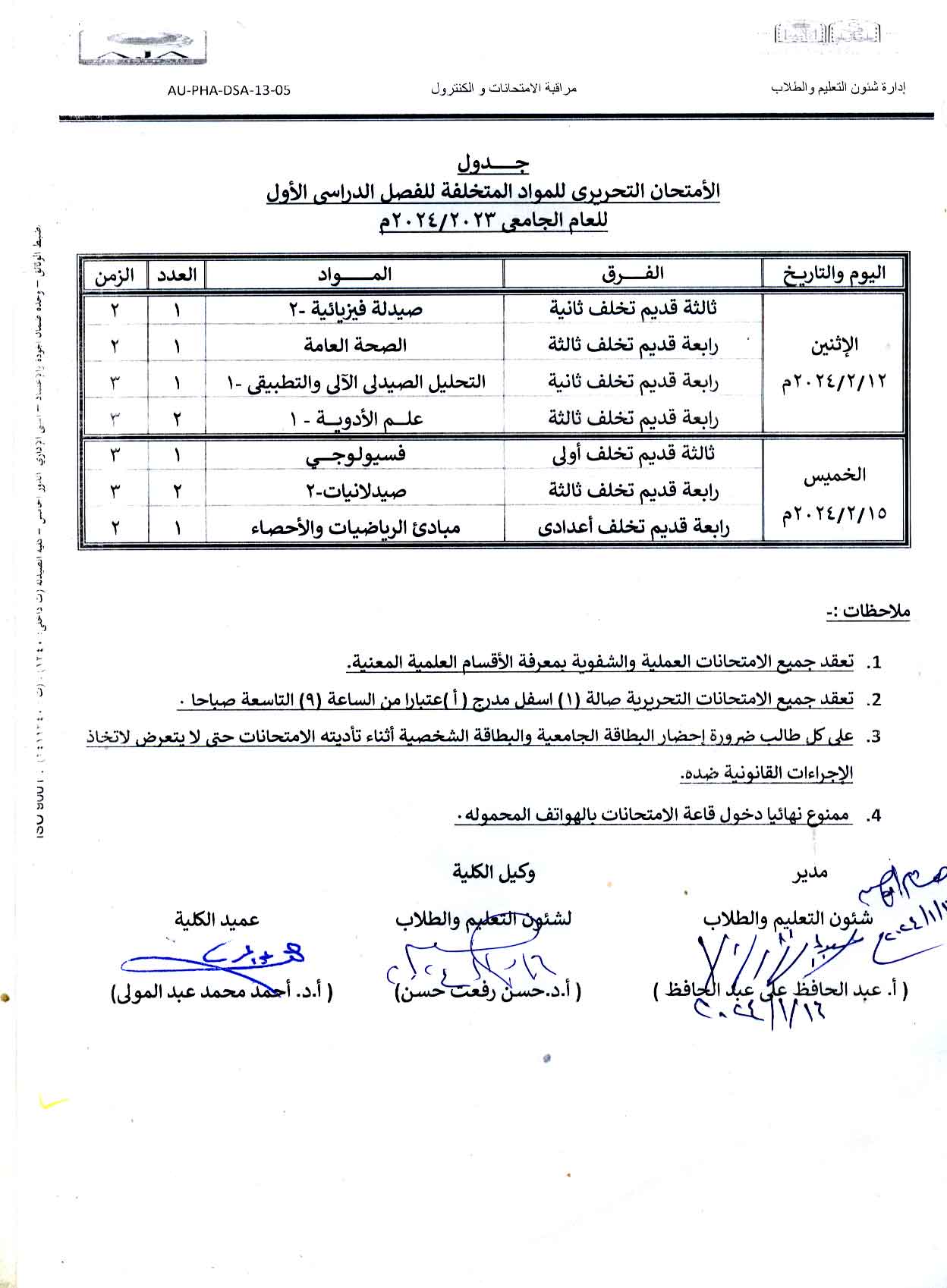
Written exam schedule for the first semester of the academic year 2023/2024


 Do you have any questions? (088) 2080369 - 2345622 Pharmacy_QAAU@pharm.aun.edu.eg
Do you have any questions? (088) 2080369 - 2345622 Pharmacy_QAAU@pharm.aun.edu.eg



The oral exam will be held on Saturday, January 20, 2024, at one o’clock in the afternoon.
Before the written exam in the pharmacognosy department according to the following committees
|
Students name |
Committee number |
|
1 Ibtihal Muhammad Mahmoud - 48 Angie Haroun Ramses |
1 |
|
49 Angie Youssef Fahim - 84- Rawan Alaa El-Din Ahmed |
2 |
|
85 Rawan Muhammad Abdel Majeed - 122 Abdel Rahman Juma Muhammad |
3 |
|
123 Abdul Rahman Omar Abdul Hamid - 161 Muhammad Ahmed Qasim |
4 |
The oral exam will be completed by holding two committees immediately after the written exam:
|
Student name |
Committee number |
|
162 Muhammad Ismail Abdel Khaleq - 202 Naglaa Rajab Ahmed |
5 |
|
203 Nada Ramadan in Deir - until the end + Fifth year students registered the course |
6 |
The announced dates must be strictly adhered to, with our sincere wishes for success
The influence of metal complexation of two polar β-lactam antibiotics was investigated using surface enhanced Raman spectroscopy (SERS) technique. SERS method was applied to track the structural changes and the degradation behaviour of the studied compounds upon Zinc (II) ions-complexation. In situ laser-induced coral reefs-like photomicroclusters have been utilized as a SERS platform. The produced coral reefs-like photomicroclusters were characterized using scanning electron microscopy (SEM) and transmission electron microscope (TEM). The antibacterial efficiency of the antibiotics was investigated and compared before and after metal complexation using two techniques; agar well diffusion and growth curve. To provide a detailed elucidation of the complexation reaction, mass fragmentation of metal- antibiotics complexes was investigated using liquid chromatography/mass spectrometric (LC/MS) technique. It was found that metal complexation of classical β-lactam antibiotic (Ticarcillin) promoted the rate of its degradation, leading to a decrease of the antibacterial efficiency. On the other side, the antibacterial activity of the newly developed β-lactam (Faropenem) has been greatly enhanced via metal-complexation reaction.
Lung cancer is one of the most aggressive and deadliest health threats. There has been an increasing interest in non-coding RNA (ncRNA) recently, especially in the areas of carcinogenesis and tumour progression. However, ncRNA-directed therapies are still encountering obstacles on their way to the clinic. In the present article, we provide an overview on the potential of targeting ncRNA in the treatment of lung cancer. Then, we discuss the delivery challenges and recent approaches enabling the delivery of ncRNA-directed therapies to the lung cancer cells, where we illuminate some advanced technologies including chemically-modified oligonucleotides, nuclear targeting, and three-dimensional in vitro models. Furthermore, advanced non-viral delivery systems recruiting nanoparticles, biomimetic delivery systems, and extracellular vesicles are also highlighted. Lastly, the challenges limiting the clinical trials on the therapeutic targeting of ncRNAs in lung cancer and future directions to tackle them are explored.
Bacterial infections caused by different strains of bacteria still one of the most important disorders affecting humans worldwide. Polymers nanocomposite systems could be considered as an alternative to conventional antibiotics to eradicate bacterial infections.
In an attempt to enhance the antibacterial performance of silver and iron oxide nanoparticles, decrease their aggregation and toxicity, a polymeric hybrid nanocomposite system combining both nanoparticles is produced.
Magnetic Ag–Fe3O4@polymer hybrid nanocomposites prepared using different polymers, namely polyethylene glycol 4000, ethyl cellulose, and chitosan were synthesized via wet impregnation and ball-milling techniques. The produced nanocomposites were tested for their physical properties and antibacterial activities.
XRD, FT-IR, VSM, and TEM results confirmed the successful preparation of hybrid nanocomposites. Hybrid nanocomposites have average crystallite sizes in the following order Ag–Fe3O4@CS (8.9 nm) < Ag–Fe3O4@EC (9.0 nm) < Ag–Fe3O4@PEG4000 (9.4 nm) and active surface area of this trend Ag–Fe3O4@CS (130.4 m2g−1) > Ag–Fe3O4@EC (128.9 m2g−1) > Ag–Fe3O4@PEG4000 (123.4 m2g−1). In addition, they have a saturation magnetization in this order: Ag–Fe3O4@PEG4000 (44.82 emu/g) > Ag–Fe3O4@EC (40.14 emu/g) > Ag–Fe3O4@CS (22.90 emu/g). Hybrid nanocomposites have a pronounced antibacterial action against Bacillus cereus, Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus intermedius compared to iron oxide nanoparticles and positive antibacterial drug. In addition, both Ag–Fe3O4@EC and Ag–Fe3O4@CS have a lower MIC values compared to Ag–Fe3O4@PEG and positive control.
Magnetic Ag–Fe3O4 hybrid nanocomposites could be promising antibacterial nanomaterials and could pave the way for the development of new materials with even more unique properties and applications.
Herein, we report on the development of chitosan-capped silver nanoparticles (AgNPs-CHI) with an intrinsic activity against breast cancer cells. Following chemical synthesis via a simple, one-pot reaction, the chitosan coating of AgNPs was verified using Fouriertransforminfraredandultraviolet–visiblespectroscopies. The physicochemical properties and morphology were characterized using dynamic light scattering, scanning electron microscopy, and transmission electron microscopy. The shelf stability of the optimized platform was tracked for 3 months upon storage at either room temperature or 4°C. Then, the anticancer activities of AgNPsCHI on human breast cancer cells, MCF-7, versus normal human cells, human skin fibroblasts (HSF), were evaluated via 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide cytotoxicity assay and tumor-associated biomarkers determination by enzyme-linked immunosorbent assay, in comparison with plain silver nitrate (AgNO3) solution. AgNPs were successfully coated with chitosan and demonstrated acceptable physicochemical properties, with a spherical morphology and high stability upon long-term storage. Although AgNPs-CHI and AgNO3 demonstrated
comparablecytotoxicitytoMCF-7cells,AgNPs-CHIresulted in 10-fold lower toxicity to HSF cells, suggesting a higher selectivity. In addition, AgNPs-CHI lowered IL-6 and tumor necrosis factor-alpha levels in MCF-7 cells by 90 and 30%, respectively, compared to 60 and 10% in the case of plain AgNO3. The interesting therapeutic modality presented in this study is promising for potential clinical applications.
Silver nanoparticles (AgNPs) have become an interesting therapeutic modality and drug delivery platform. Herein, we aimed to investigate the impact of functional coating on the in vivo performance of AgNPs as an economic and scalable method to modulate their behavior.
AgNPs were coated with chitosan (CHI) as a model biopolymer using a one-pot reduction-based method, where CHI of two molecular weight ranges were investigated. The resultant CHI-coated AgNPs (AgNPs-CHI) were characterized using UV-VIS spectroscopy, DLS, and TEM. AgNPs were administered intravenously to rats and their biodistribution and serum levels of hepato-renal function markers were monitored 24 h later compared to plain AgNO3 as a positive control.
UV-VIS spectroscopy confirmed the successful coating of AgNPs with CHI. DLS revealed the superiority of medium molecular weight CHI over its low molecular weight counterpart. AgNPs-CHI demonstrated a semi-complete clearance from the systemic circulation, a liver-dominated tissue tropism, and limited renal exposure. On the other hand, AgNO3 was poorly cleared from the circulation, with relatively high renal exposure and a non-specific tissue tropism. AgNPs-CHI were well-tolerated by the liver and kidney without signs of toxicity or inflammation, in contrary with AgNO3 which resulted in a significant elevation of Creatinine (CRE), Urea, and Total Protein (TP), suggesting a significant nephrotoxicity and inflammation.
Functional coating of AgNPs with CHI substantially modulated their in vivo behavior, promoting their hepatic selectivity and biotolerability, which can be invested in the development of drug delivery systems for the treatment of liver diseases.

