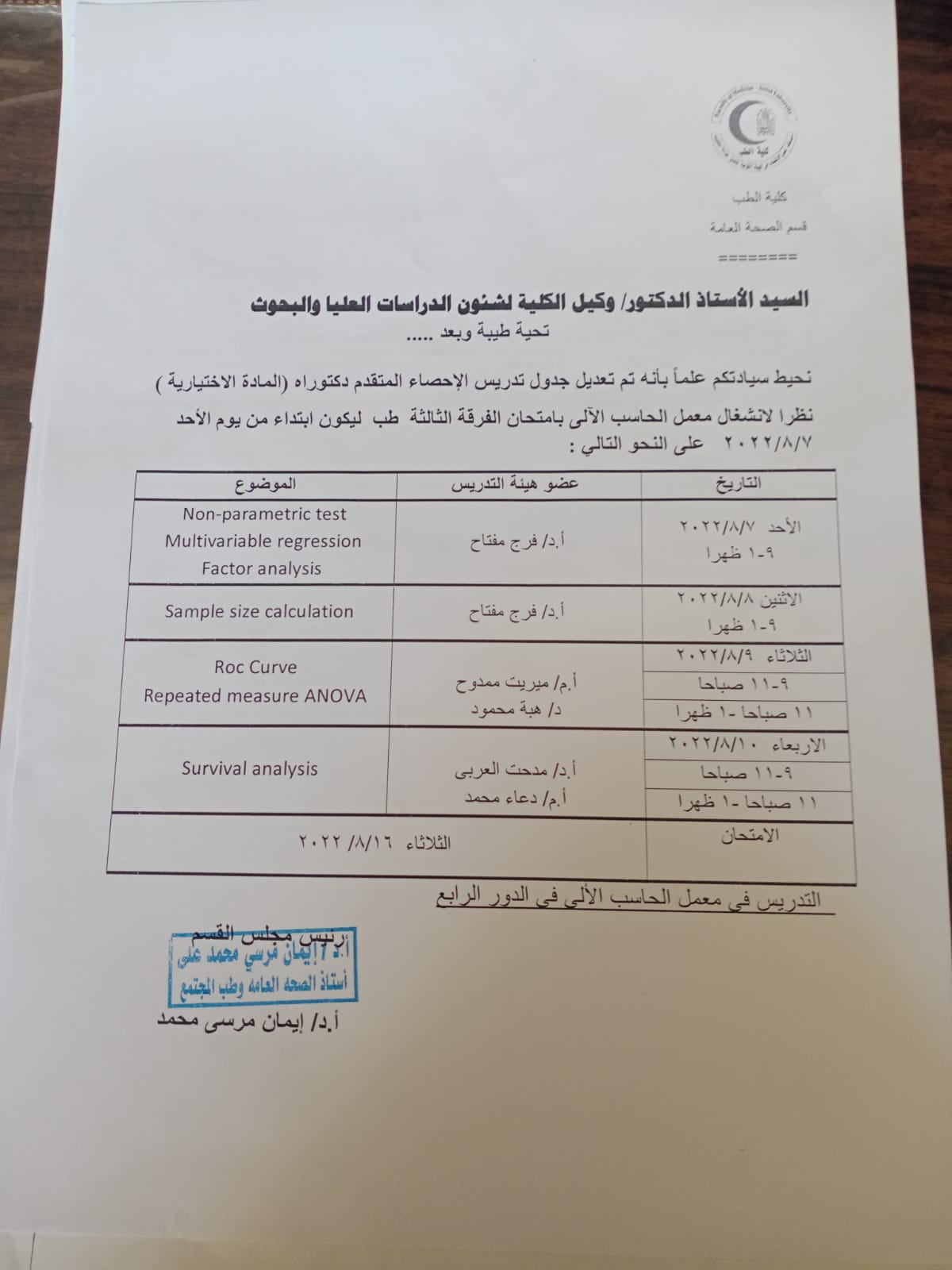
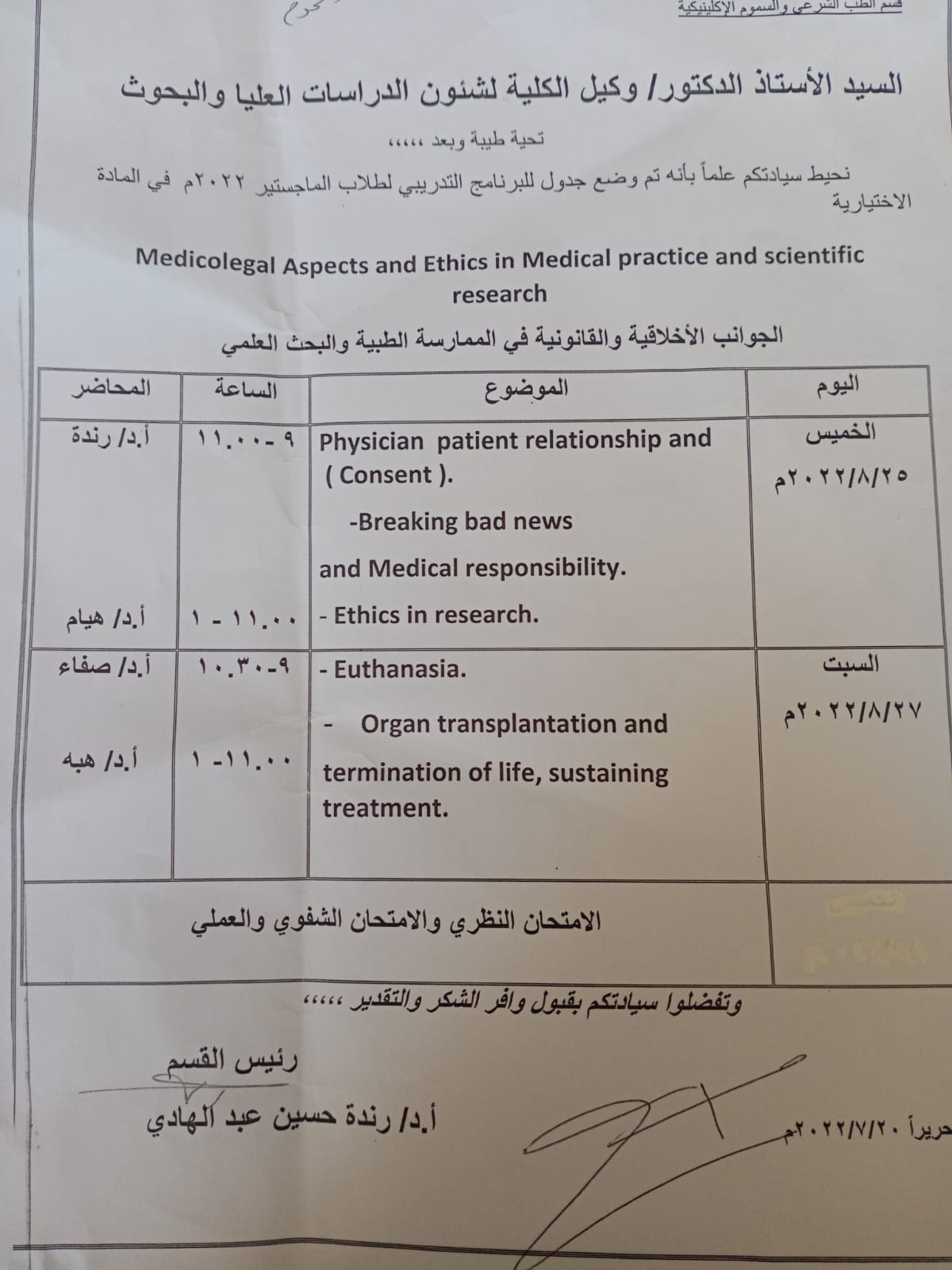
The activities of the DNA lab course for forensic medicine and diagnosis
تحت رعاية
ا. د/طارق الجمال رئيس الجامعه
ا. د/ علاء عطيه عميد الكلية ورئيس مجلس إدارة المستشفيات الجامعية
ا.د/سعد زكي وكيل الكلية لشئون البيئة وخدمة المجتمع
ورئاسة
ا. د/ رنده حسين رئيس قسم الطب الشرعي والسموم الاكلينيكية
ا.د/هاله فتحي مدير معمل الحمض النووي
ا. د/ هبه عطيه يسي. مقرر الدورة
انتهاء فعاليات دورة معمل الحمض النووي للطب
الشرعي و التشخيص بنجاح
والتي تم عقدها في الفتره من ٢٧ إلي ٢٨ يوليو ٢٠٢٢
حضر الدورة التدريبية أطباء من كلية طب المنيا وكلية الطب باسوان
ودارت حول أهمية فحص الحمض النووي وكيفية استخلاصه وتحليله ومطابقته بقضايا الطب الشرعي
 Do you have any questions?
Do you have any questions?