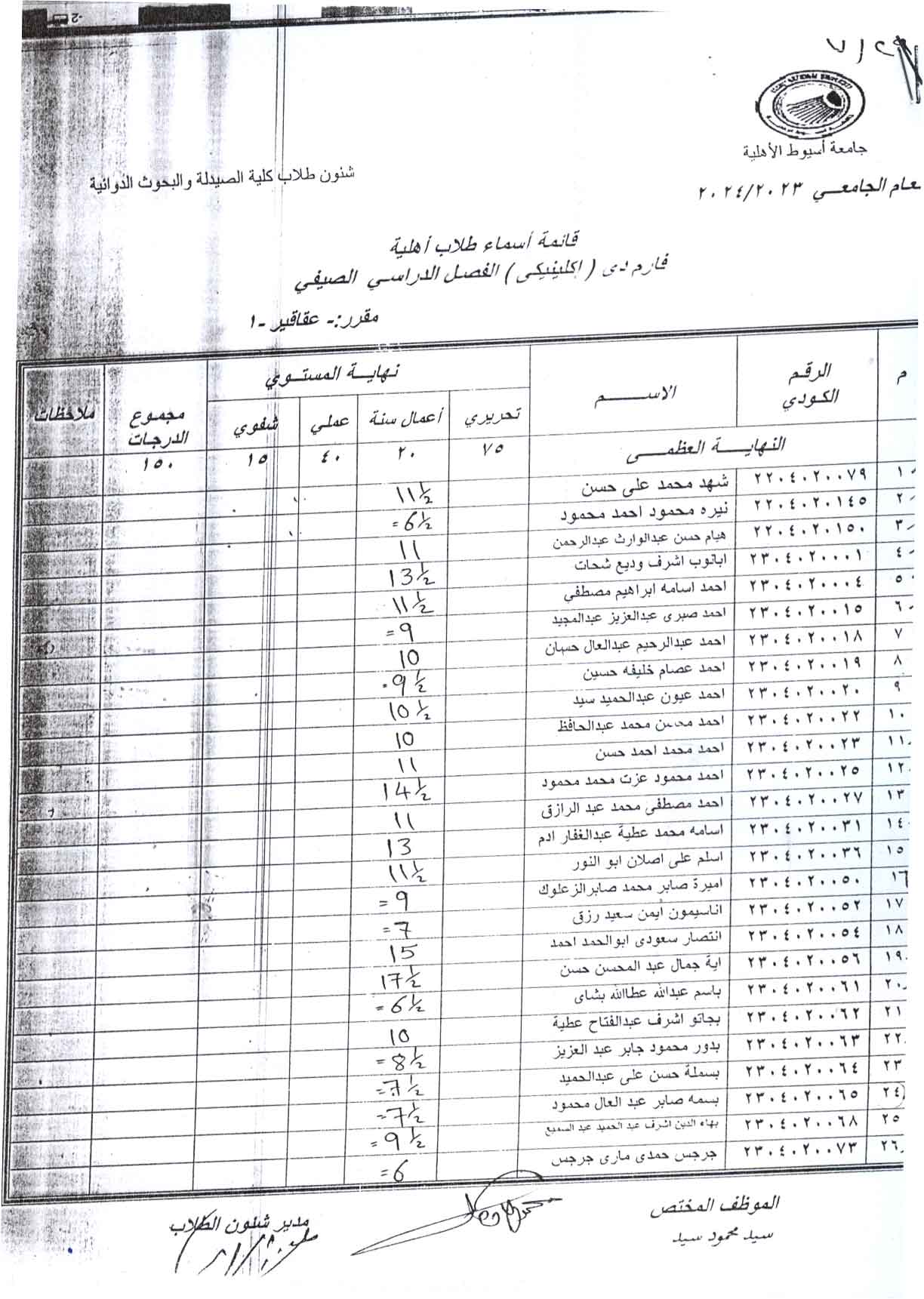
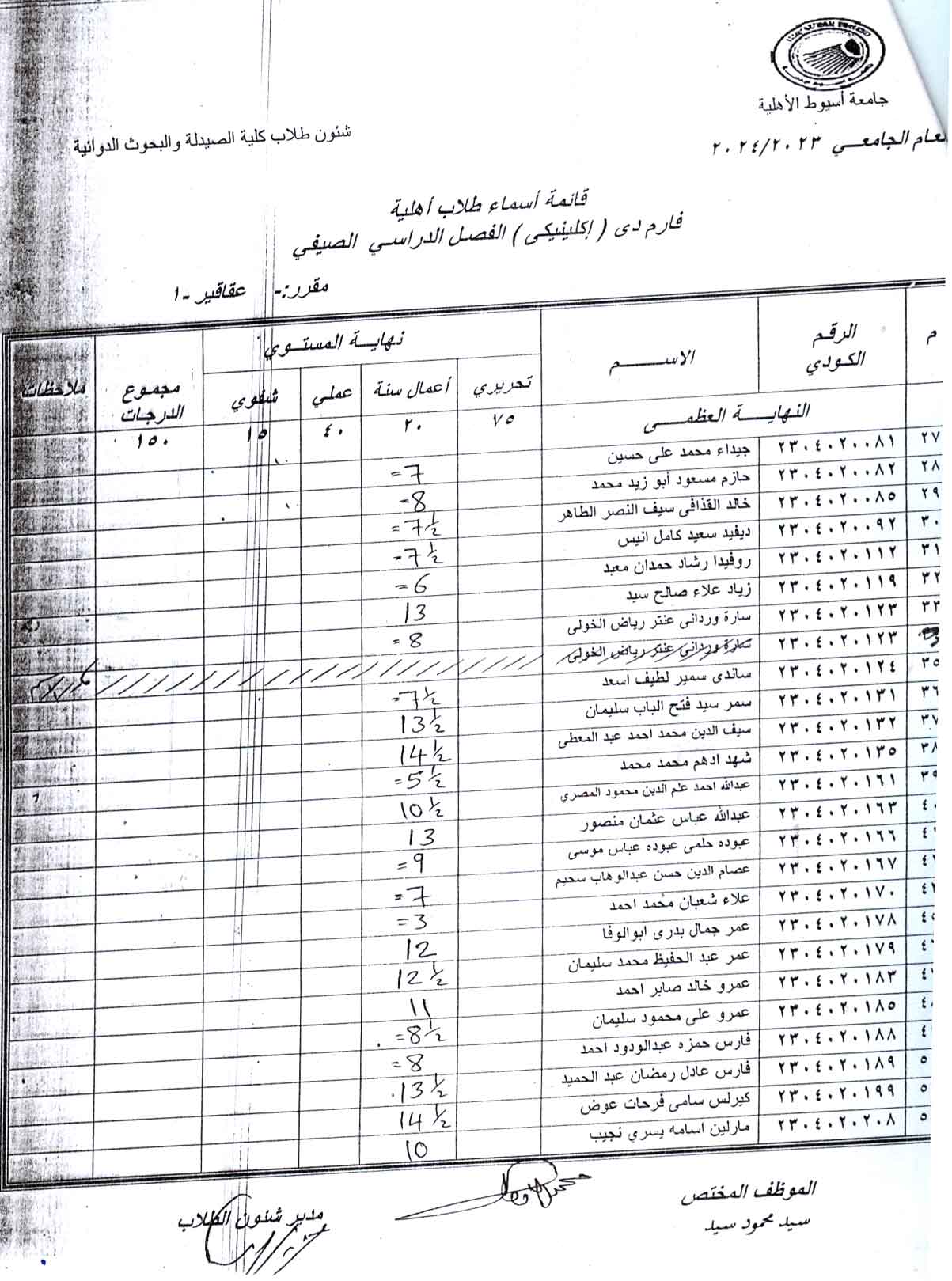
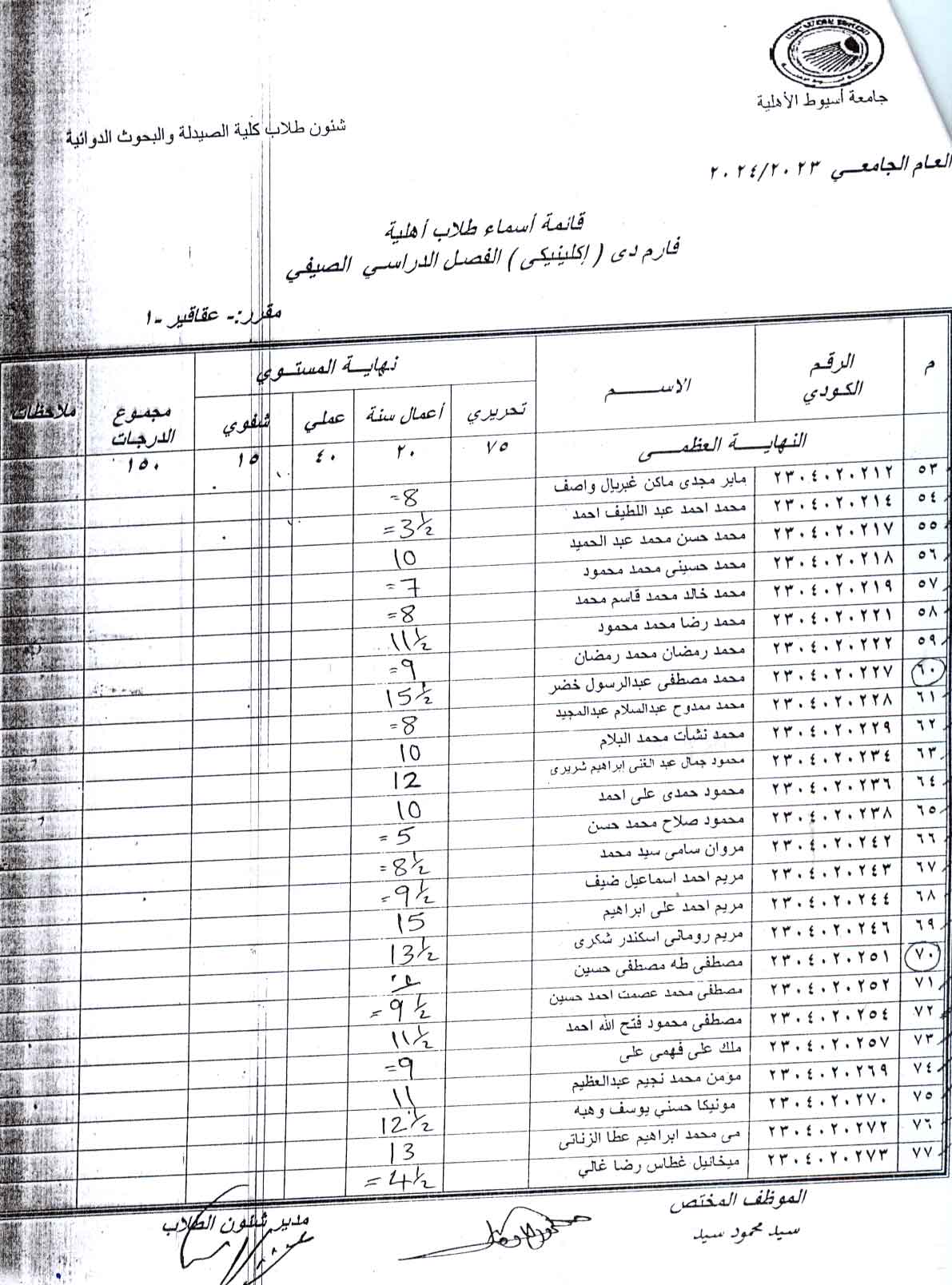
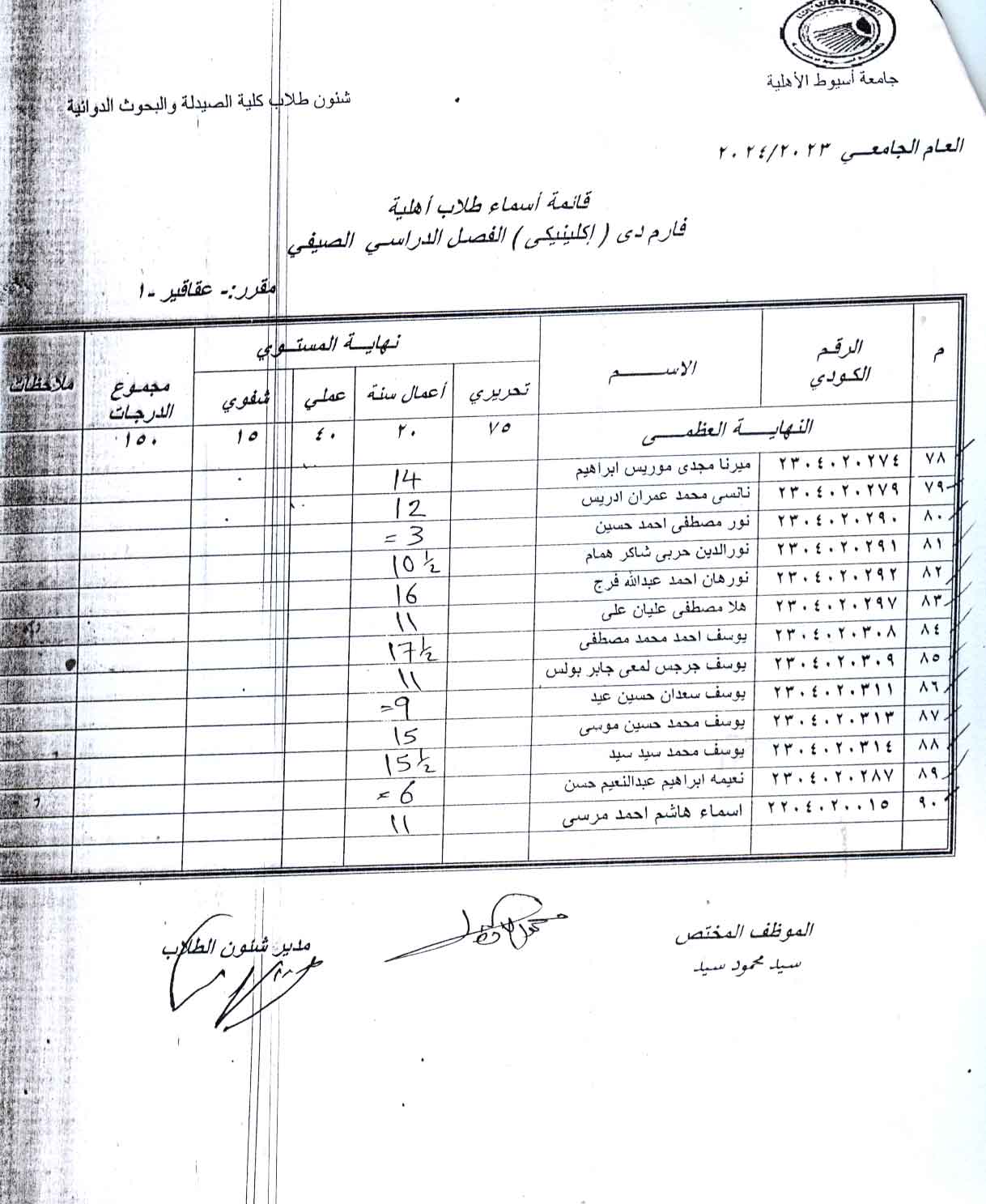
Adenine model of chronic renal failure in rats to determine whether MCC950, an NLRP3 inflammasome inhibitor, is a renopreventive
Background
Chronic renal failure (CRF) is defined by a significant decline in renal function that results in decreased salt filtration and inhibition of tubular reabsorption, which ultimately causes volume enlargement. This study evaluated the potential renopreventive effects of the NLRP3 inflammasome inhibitor MCC950 in adenine-induced CRF in rats due to conflicting evidence on the effects of MCC950 on the kidney.
Methods
Since the majority of the kidney tubular abnormalities identified in people with chronic renal disease are comparable to those caused by adding 0.75 percent of adenine powder to a rat's diet each day for four weeks, this method has received broad approval as a model for evaluating kidney damage. Throughout the test, blood pressure was checked weekly and at the beginning. Additionally, oxidative stress factors, urine sample examination, histological modifications, and immunohistochemical adjustments of caspase-3 and interleukin-1 beta (IL-1) levels in renal tissues were carried out.
Results
Results revealed that MCC950, an inhibitor of the NLRP3 inflammasome, had a renopreventive effect, which was demonstrated by a reduction in blood pressure readings and an improvement in urine, serum, and renal tissue indicators that indicate organ damage. This was also demonstrated by the decrease in neutrophil gelatinase-associated lipocalin tubular expression (NGAL).
The NLRP3 inflammasome inhibitor MCC950 was found to significantly alleviate the worsening renal cellular alterations evidenced by increased expression of caspase-3 and IL-1, according to immunohistochemical tests.
Conclusion
The NLRP3 inflammasome inhibitor MCC950 demonstrated renopreventive effects in the CRF rat model, suggesting that it might be used as a treatment strategy to stop the progression of CRF.