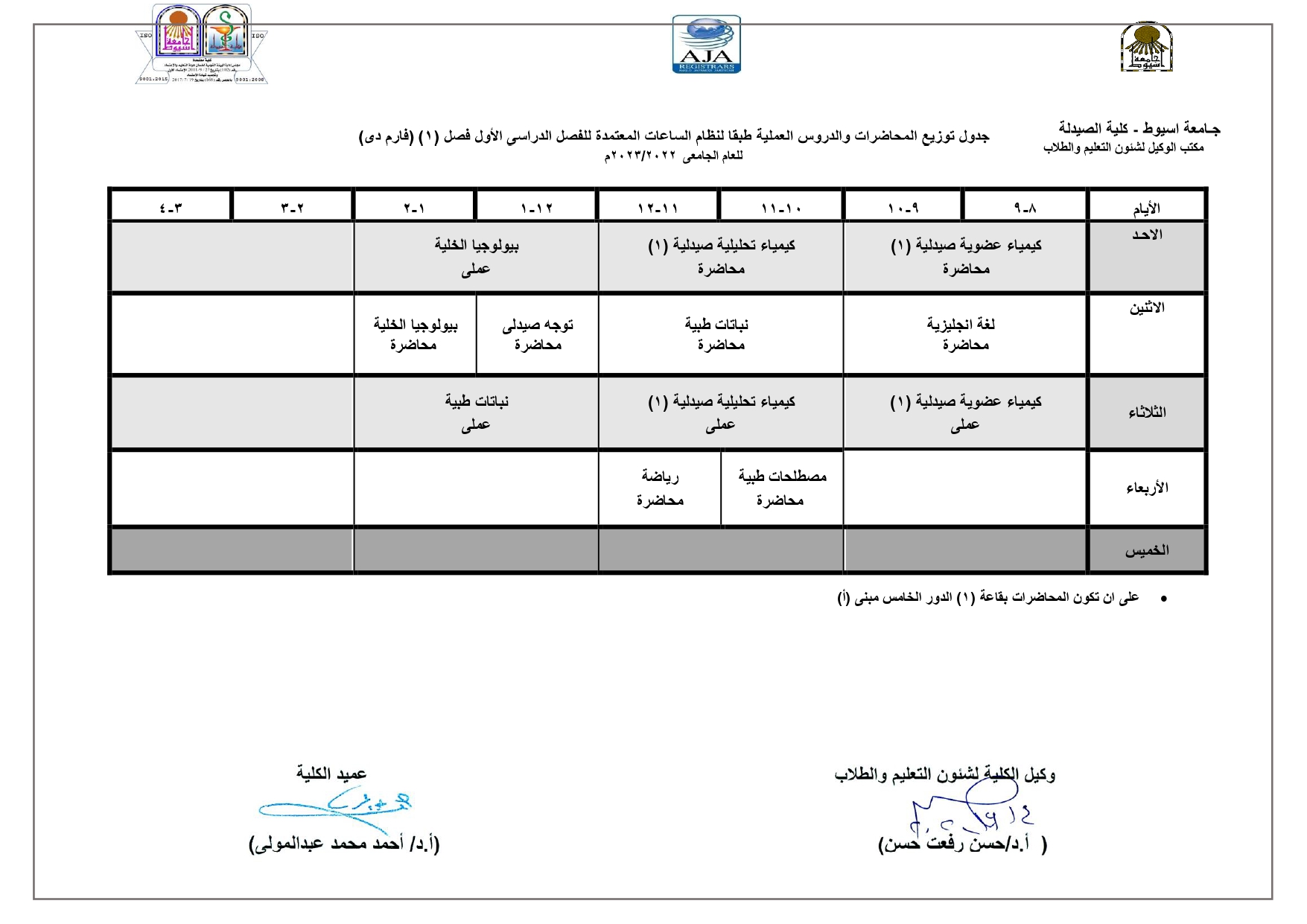
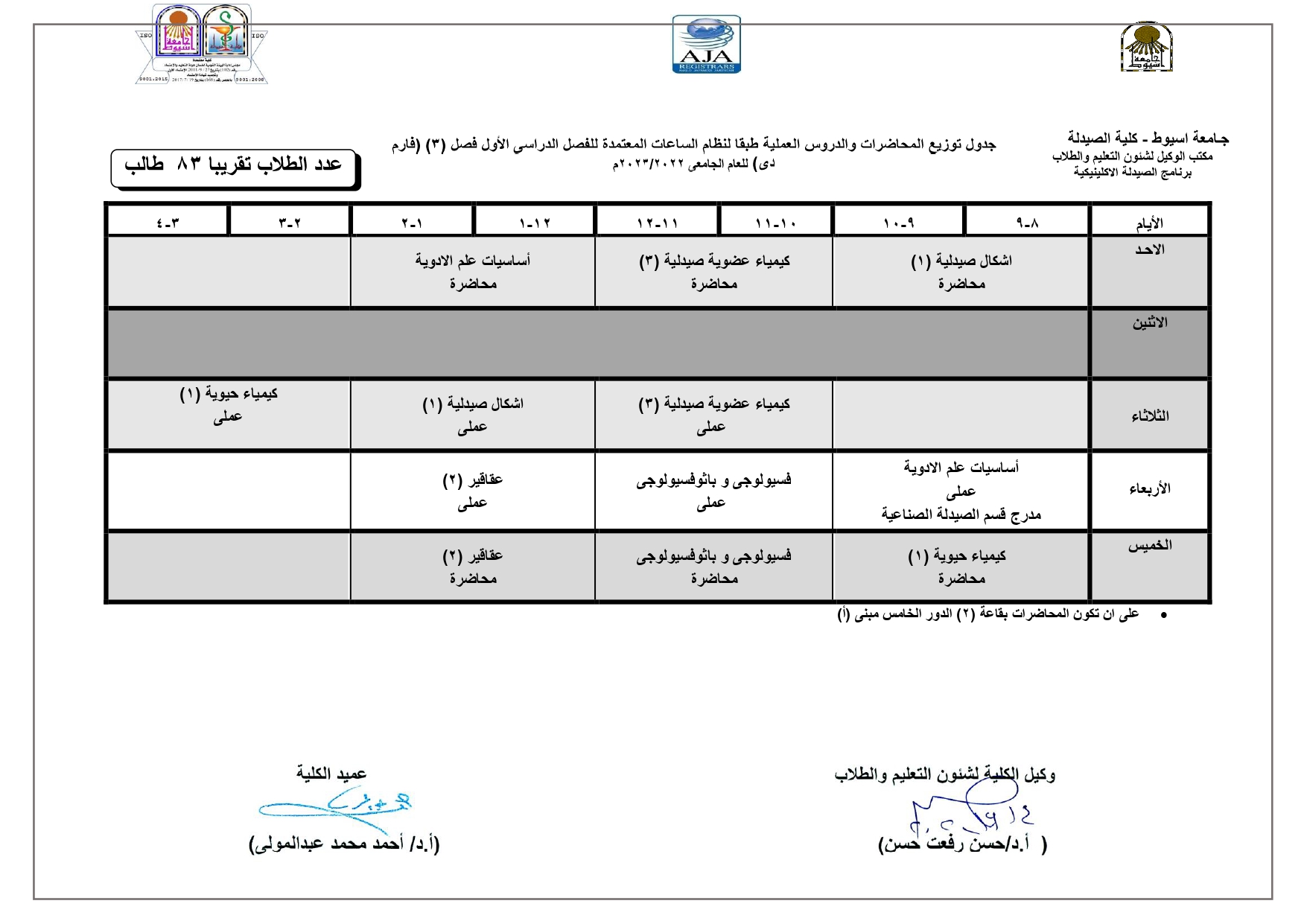
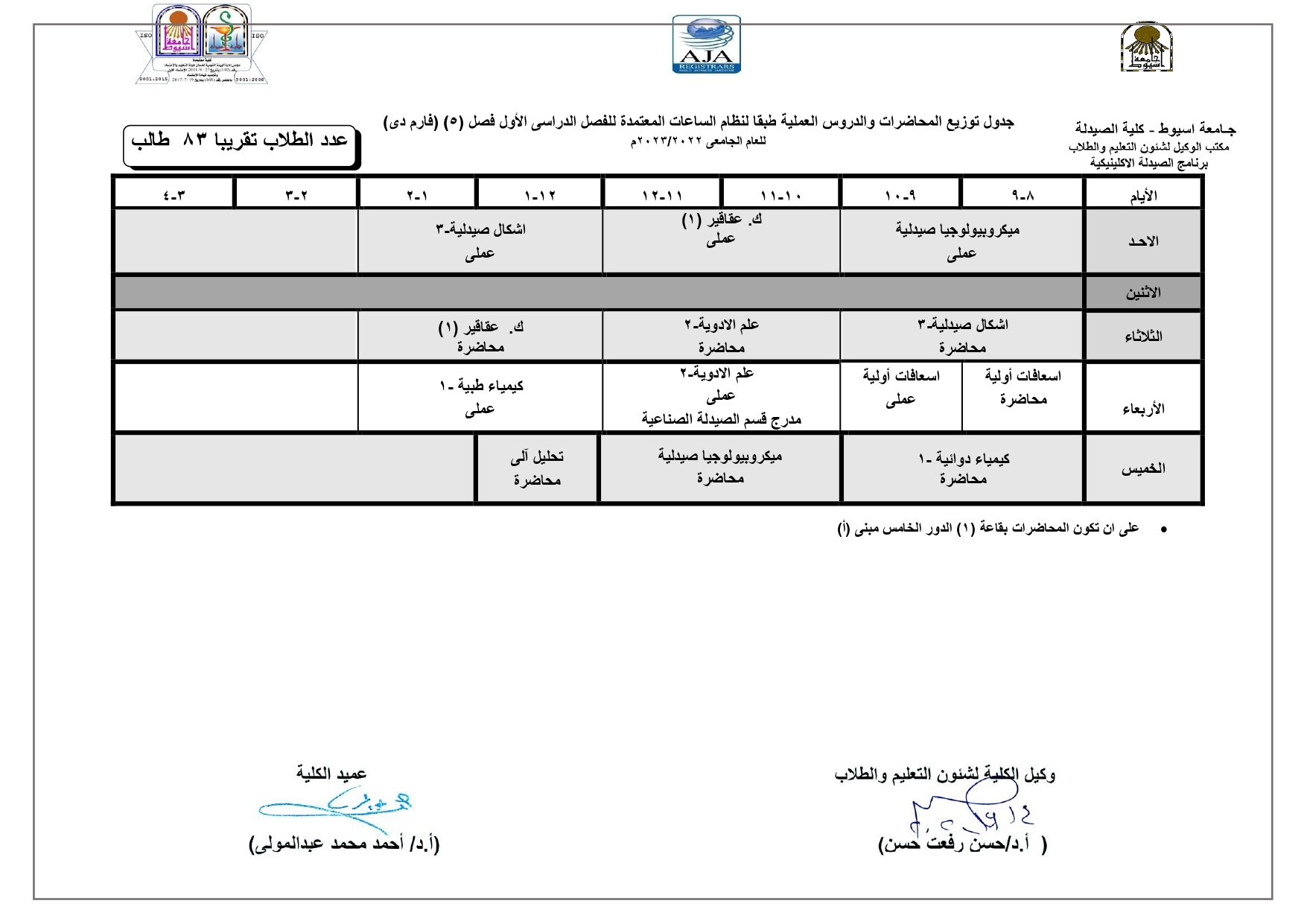
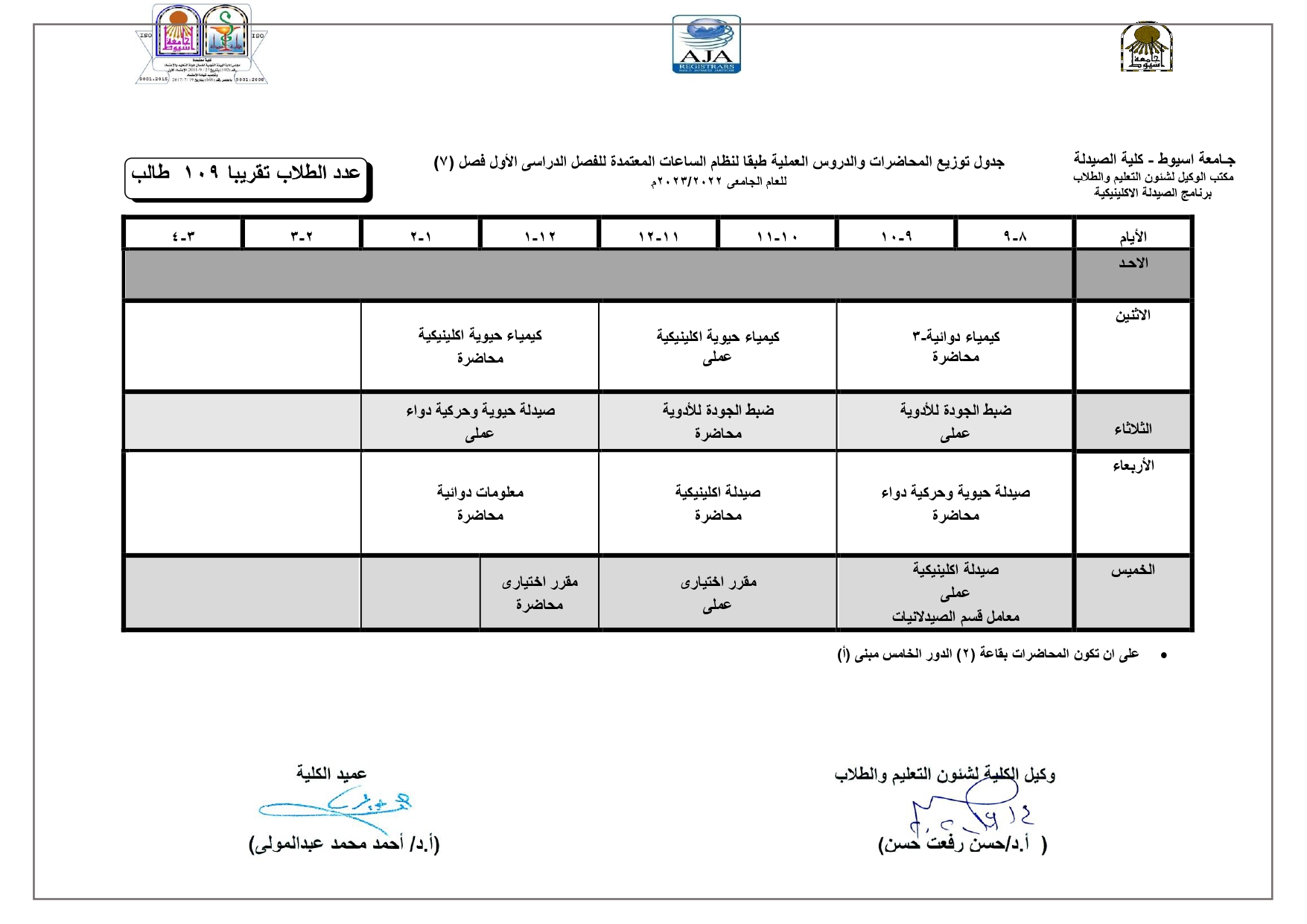
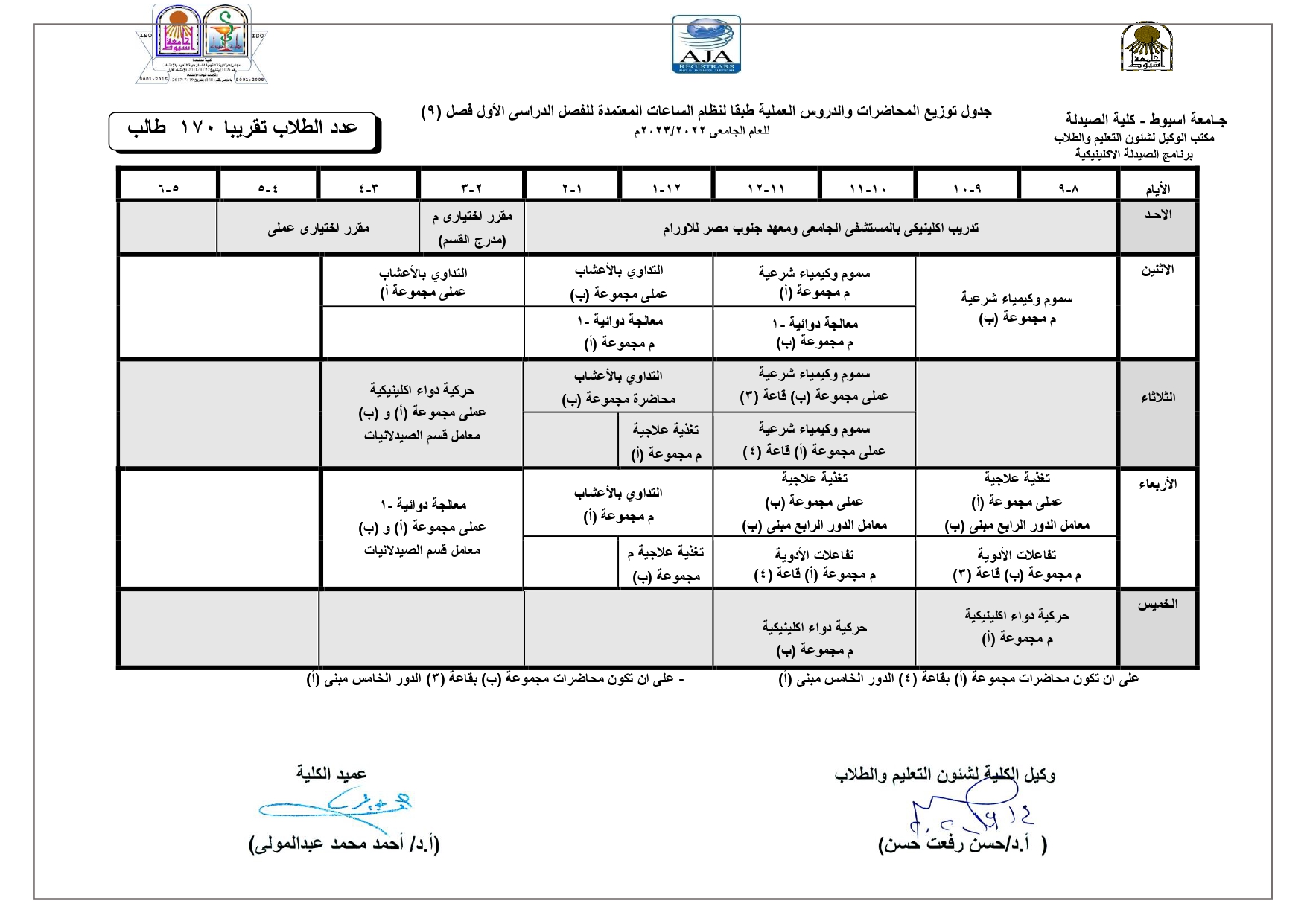
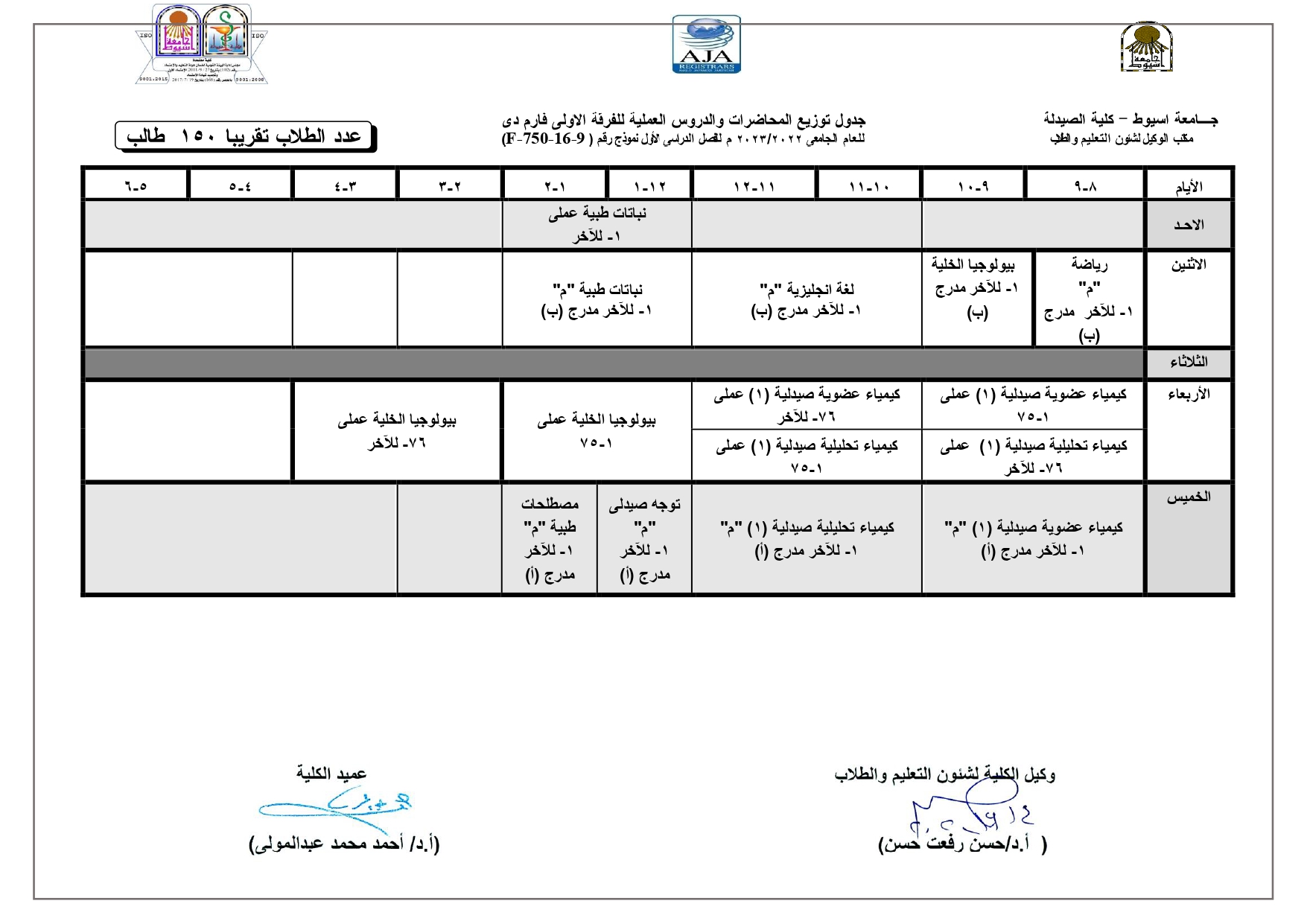
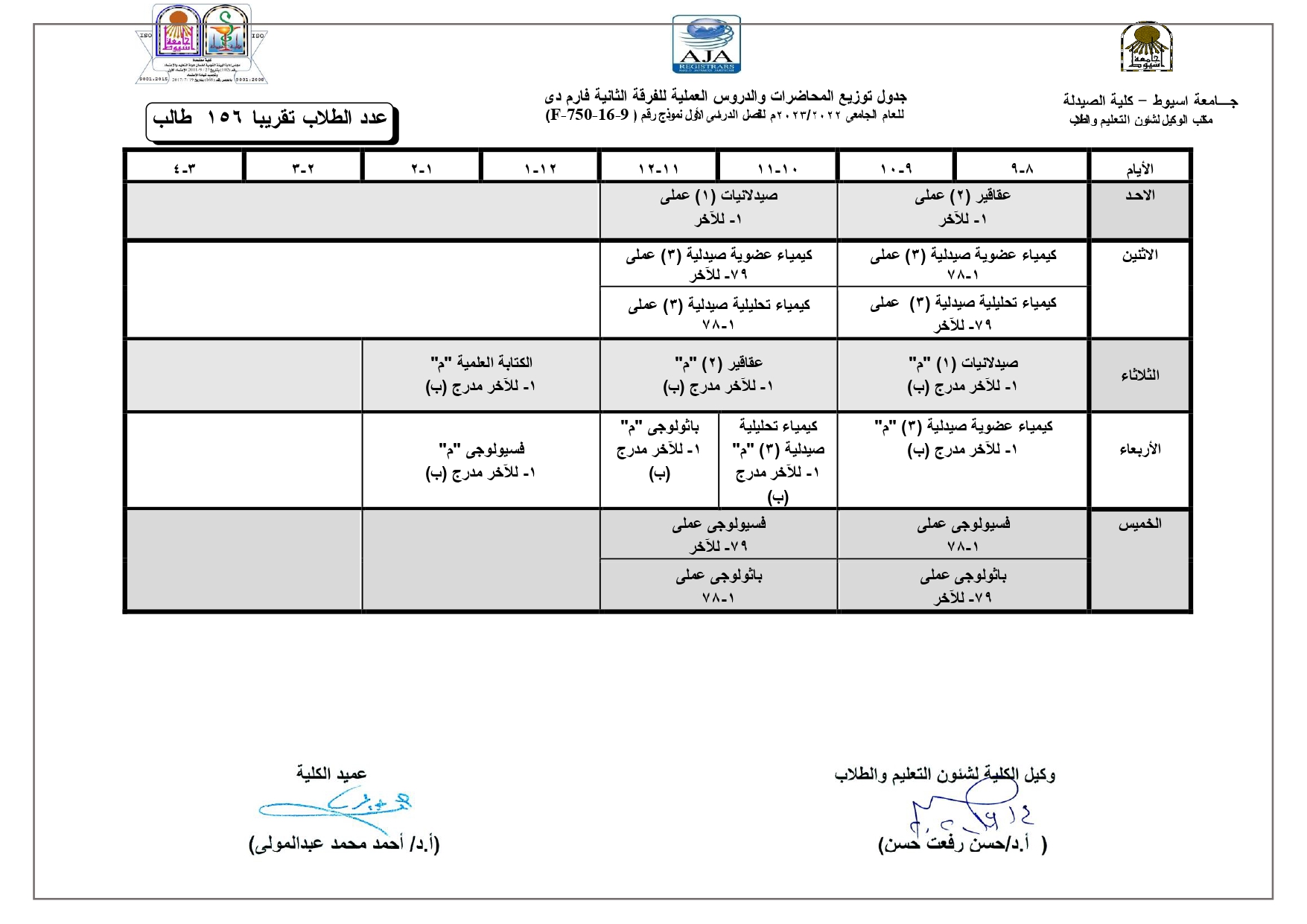
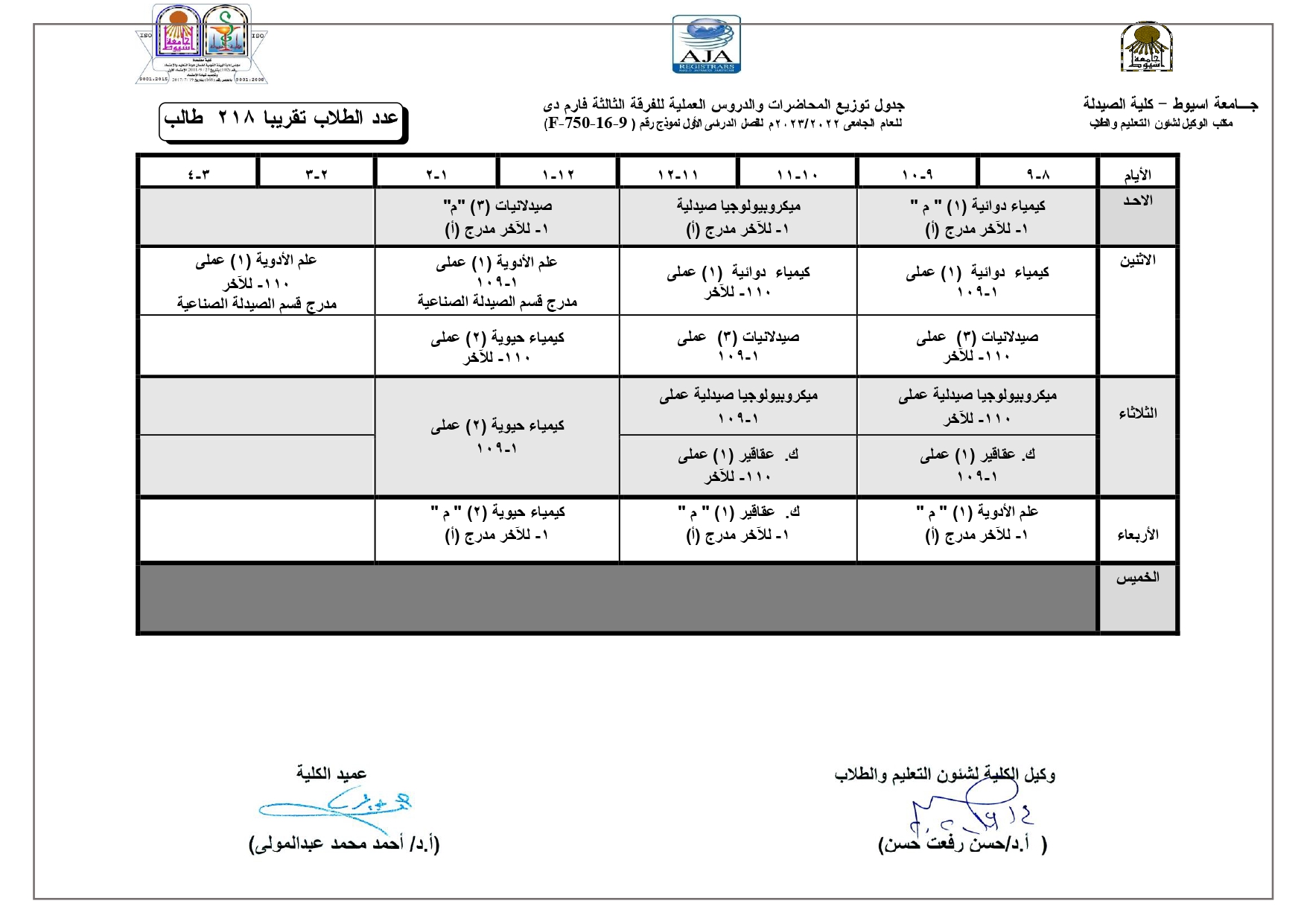
Elegant, Flexible Vesicular Nanocarriers for the Efficient Skin Delivery of Topically Applied Drugs
vesicles have shown tremendous potential to overcome these hurdles and improve the local therapeutic effect of these drugs.
Objective: This review article is aimed to shed light on flexible nano-vesicular carriers as a means to combat skin diseases.
Methods: The literature was reviewed using PubMed database using various keywords such as liposomes, flexible (deformable liposomes) (transferosomes), ethosomes, transethosomes, niosomes, and spanlastics.
Results: Liposomes and niosomes were found effective for the loading and release of both hydrophilic and lipophilic drugs. However, their limited skin penetration led to drug delivery to the outermost layers of skin only. This necessitates the search for innovative vesicular carriers, including liposomes, flexible (deformable liposomes), ethosomes, transethosomes, and spanlastics. These flexible nano-vesicular carriers showed enhanced drug delivery and deposition across various skin layers, which was better than their corresponding conventional vesicles. This resulted in superior drug efficacy against various skin diseases such as skin cancer, inflammatory skin diseases, superficial fungal infections, etc.
Conclusion: Flexible nano-vesicular carriers have proven themselves as efficient drug delivery systems that are able to deliver their cargo into the deep skin layers and thus, improve the therapeutic outcome of various skin diseases. However, there remain some challenges that need to be addressed before these nanocarriers can be translated from the lab to clinics.